+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-22423 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of an endocytic receptor | |||||||||
 Map data Map data | half1 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Cell-surface receptor / Immune receptor / mannose receptor family / IMMUNE SYSTEM | |||||||||
| Function / homology |  Function and homology information Function and homology informationendocytosis / signaling receptor activity / carbohydrate binding / immune response / inflammatory response / external side of plasma membrane / extracellular exosome / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
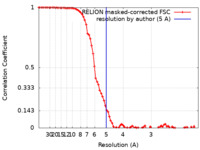
| Method | single particle reconstruction / cryo EM / Resolution: 5.0 Å | |||||||||
 Authors Authors | Gully BS / Rossjohn J | |||||||||
| Funding support |  Australia, 2 items Australia, 2 items
| |||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2021 Journal: J Biol Chem / Year: 2021Title: The cryo-EM structure of the endocytic receptor DEC-205. Authors: Benjamin S Gully / Hariprasad Venugopal / Alex J Fulcher / Zhihui Fu / Jessica Li / Felix A Deuss / Carmen Llerena / William R Heath / Mireille H Lahoud / Irina Caminschi / Jamie Rossjohn / Richard Berry /   Abstract: DEC-205 (CD205), a member of the macrophage mannose receptor protein family, is the prototypic endocytic receptor of dendritic cells, whose ligands include phosphorothioated cytosine-guanosine ...DEC-205 (CD205), a member of the macrophage mannose receptor protein family, is the prototypic endocytic receptor of dendritic cells, whose ligands include phosphorothioated cytosine-guanosine oligonucleotides, a motif often seen in bacterial or viral DNA. However, despite growing biological and clinical significance, little is known about the structural arrangement of this receptor or any of its family members. Here, we describe the 3.2 Å cryo-EM structure of human DEC-205, thereby illuminating the structure of the mannose receptor protein family. The DEC-205 monomer forms a compact structure comprising two intercalated rings of C-type lectin-like domains, where the N-terminal cysteine-rich and fibronectin domains reside at the central intersection. We establish a pH-dependent oligomerization pathway forming tetrameric DEC-205 using solution-based techniques and ultimately solved the 4.9 Å cryo-EM structure of the DEC-205 tetramer to identify the unfurling of the second lectin ring which enables tetramer formation. Furthermore, we suggest the relevance of this oligomerization pathway within a cellular setting, whereby cytosine-guanosine binding appeared to disrupt this cell-surface oligomer. Accordingly, we provide insight into the structure and oligomeric assembly of the DEC-205 receptor. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_22423.map.gz emd_22423.map.gz | 8.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-22423-v30.xml emd-22423-v30.xml emd-22423.xml emd-22423.xml | 19.3 KB 19.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_22423_fsc.xml emd_22423_fsc.xml | 8.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_22423.png emd_22423.png | 152.1 KB | ||
| Masks |  emd_22423_msk_1.map emd_22423_msk_1.map | 48.9 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-22423.cif.gz emd-22423.cif.gz | 6.9 KB | ||
| Others |  emd_22423_half_map_1.map.gz emd_22423_half_map_1.map.gz emd_22423_half_map_2.map.gz emd_22423_half_map_2.map.gz | 36.3 MB 36.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-22423 http://ftp.pdbj.org/pub/emdb/structures/EMD-22423 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22423 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22423 | HTTPS FTP |
-Related structure data
| Related structure data |  7jpuMC  7jptC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_22423.map.gz / Format: CCP4 / Size: 48.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_22423.map.gz / Format: CCP4 / Size: 48.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
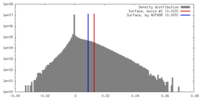
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
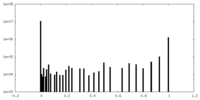
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_22423_msk_1.map emd_22423_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
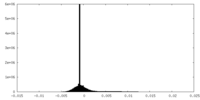
| Density Histograms |
-Half map: half2
| File | emd_22423_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
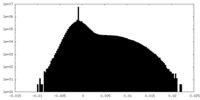
| Density Histograms |
-Half map: half2
| File | emd_22423_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Lymphocyte antigen 75, DEC205, CD205
| Entire | Name: Lymphocyte antigen 75, DEC205, CD205 |
|---|---|
| Components |
|
-Supramolecule #1: Lymphocyte antigen 75, DEC205, CD205
| Supramolecule | Name: Lymphocyte antigen 75, DEC205, CD205 / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 200 KDa |
-Macromolecule #1: Lymphocyte antigen 75
| Macromolecule | Name: Lymphocyte antigen 75 / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 198.553172 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MRTGWATPRR PAGLLMLLFW FFDLAEPSGR AANDPFTIVH GNTGKCIKPV YGWIVADDCD ETEDKLWKWV SQHRLFHLHS QKCLGLDIT KSVNELRMFS CDSSAMLWWK CEHHSLYGAA RYRLALKDGH GTAISNASDV WKKGGSEESL CDQPYHEIYT R DGNSYGRP ...String: MRTGWATPRR PAGLLMLLFW FFDLAEPSGR AANDPFTIVH GNTGKCIKPV YGWIVADDCD ETEDKLWKWV SQHRLFHLHS QKCLGLDIT KSVNELRMFS CDSSAMLWWK CEHHSLYGAA RYRLALKDGH GTAISNASDV WKKGGSEESL CDQPYHEIYT R DGNSYGRP CEFPFLIDGT WHHDCILDED HSGPWCATTL NYEYDRKWGI CLKPENGCED NWEKNEQFGS CYQFNTQTAL SW KEAYVSC QNQGADLLSI NSAAELTYLK EKEGIAKIFW IGLNQLYSAR GWEWSDHKPL NFLNWDPDRP SAPTIGGSSC ARM DAESGL WQSFSCEAQL PYVCRKPLNN TVELTDVWTY SDTRCDAGWL PNNGFCYLLV NESNSWDKAH AKCKAFSSDL ISIH SLADV EVVVTKLHNE DIKEEVWIGL KNINIPTLFQ WSDGTEVTLT YWDENEPNVP YNKTPNCVSY LGELGQWKVQ SCEEK LKYV CKRKGEKLND ASSDKMCPPD EGWKRHGETC YKIYEDEVPF GTNCNLTITS RFEQEYLNDL MKKYDKSLRK YFWTGL RDV DSCGEYNWAT VGGRRRAVTF SNWNFLEPAS PGGCVAMSTG KSVGKWEVKD CRSFKALSIC KKMSGPLGPE EASPKPD DP CPEGWQSFPA SLSCYKVFHA ERIVRKRNWE EAERFCQALG AHLSSFSHVD EIKEFLHFLT DQFSGQHWLW IGLNKRSP D LQGSWQWSDR TPVSTIIMPN EFQQDYDIRD CAAVKVFHRP WRRGWHFYDD REFIYLRPFA CDTKLEWVCQ IPKGRTPKT PDWYNPDRAG IHGPPLIIEG SEYWFVADLH LNYEEAVLYC ASNHSFLATI TSFVGLKAIK NKIANISGDG QKWWIRISEW PIDDHFTYS RYPWHRFPVT FGEECLYMSA KTWLIDLGKP TDCSTKLPFI CEKYNVSSLE KYSPDSAAKV QCSEQWIPFQ N KCFLKIKP VSLTFSQASD TCHSYGGTLP SVLSQIEQDF ITSLLPDMEA TLWIGLRWTA YEKINKWTDN RELTYSNFHP LL VSGRLRI PENFFEEESR YHCALILNLQ KSPFTGTWNF TSCSERHFVS LCQKYSEVKS RQTLQNASET VKYLNNLYKI IPK TLTWHS AKRECLKSNM QLVSITDPYQ QAFLSVQALL HNSSLWIGLF SQDDELNFGW SDGKRLHFSR WAETNGQLED CVVL DTDGF WKTVDCNDNQ PGAICYYSGN ETEKEVKPVD SVKCPSPVLN TPWIPFQNCC YNFIITKNRH MATTQDEVHT KCQKL NPKS HILSIRDEKE NNFVLEQLLY FNYMASWVML GITYRNKSLM WFDKTPLSYT HWRAGRPTIK NEKFLAGLST DGFWDI QTF KVIEEAVYFH QHSILACKIE MVDYKEEYNT TLPQFMPYED GIYSVIQKKV TWYEALNMCS QSGGHLASVH NQNGQLF LE DIVKRDGFPL WVGLSSHDGS ESSFEWSDGS TFDYIPWKGQ TSPGNCVLLD PKGTWKHEKC NSVKDGAICY KPTKSKKL S RLTYSSRCPA AKENGSRWIQ YKGHCYKSDQ ALHSFSEAKK LCSKHDHSAT IVSIKDEDEN KFVSRLMREN NNITMRVWL GLSQHSVDQS WSWLDGSEVT FVKWENKSKS GVGRCSMLIA SNETWKKVEC EHGFGRVVCK VPLGPDYTAI AIIVATLSIL VLMGGLIWF LFQRHRLHLA GFSSVRYAQG VNEDEIMLPS FHD UniProtKB: Lymphocyte antigen 75 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.1 mg/mL | ||||||
|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
Details: 20 mM Tris-HCl at pH 8.0, 150 mM NaCl buffer | ||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER | ||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK II | ||||||
| Details | 3 microlitres of sample in 20 mM Tris-HCl at pH 8.0, 150 mM NaCl buffer |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 63.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT | ||||||||
| Output model |  PDB-7jpu: |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)