+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Complex of ASCT2 with Suppressyn | |||||||||
 Map data Map data | Main map from 3D flex reconstruction in Cryosparc. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Small neural amino acid transporter / ASCT2 / Receptor binding domain / Suppressyn / PROTEIN TRANSPORT | |||||||||
| Function / homology | : / : / Suppressyn Function and homology information Function and homology information | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.39 Å | |||||||||
 Authors Authors | Khare S / Kumar A / Reyes N | |||||||||
| Funding support | European Union, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Receptor-recognition and antiviral mechanisms of retrovirus-derived human proteins. Authors: Shashank Khare / Miryam I Villalba / Juan C Canul-Tec / Arantza Balsebre Cajiao / Anand Kumar / Marija Backovic / Felix A Rey / Els Pardon / Jan Steyaert / Camilo Perez / Nicolas Reyes /     Abstract: Human syncytin-1 and suppressyn are cellular proteins of retroviral origin involved in cell-cell fusion events to establish the maternal-fetal interface in the placenta. In cell culture, they ...Human syncytin-1 and suppressyn are cellular proteins of retroviral origin involved in cell-cell fusion events to establish the maternal-fetal interface in the placenta. In cell culture, they restrict infections from members of the largest interference group of vertebrate retroviruses, and are regarded as host immunity factors expressed during development. At the core of the syncytin-1 and suppressyn functions are poorly understood mechanisms to recognize a common cellular receptor, the membrane transporter ASCT2. Here, we present cryo-electron microscopy structures of human ASCT2 in complexes with the receptor-binding domains of syncytin-1 and suppressyn. Despite their evolutionary divergence, the two placental proteins occupy similar positions in ASCT2, and are stabilized by the formation of a hybrid β-sheet or 'clamp' with the receptor. Structural predictions of the receptor-binding domains of extant retroviruses indicate overlapping binding interfaces and clamping sites with ASCT2, revealing a competition mechanism between the placental proteins and the retroviruses. Our work uncovers a common ASCT2 recognition mechanism by a large group of endogenous and disease-causing retroviruses, and provides high-resolution views on how placental human proteins exert morphological and immunological functions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17193.map.gz emd_17193.map.gz | 32.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17193-v30.xml emd-17193-v30.xml emd-17193.xml emd-17193.xml | 23.9 KB 23.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17193_fsc.xml emd_17193_fsc.xml | 8.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_17193.png emd_17193.png | 84.4 KB | ||
| Filedesc metadata |  emd-17193.cif.gz emd-17193.cif.gz | 6.7 KB | ||
| Others |  emd_17193_additional_1.map.gz emd_17193_additional_1.map.gz emd_17193_additional_2.map.gz emd_17193_additional_2.map.gz emd_17193_half_map_1.map.gz emd_17193_half_map_1.map.gz emd_17193_half_map_2.map.gz emd_17193_half_map_2.map.gz | 4.8 MB 4.8 MB 226.8 MB 226.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17193 http://ftp.pdbj.org/pub/emdb/structures/EMD-17193 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17193 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17193 | HTTPS FTP |
-Related structure data
| Related structure data |  8ouiMC  8oudC  8ouhC  8oujC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_17193.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17193.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Main map from 3D flex reconstruction in Cryosparc. | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.814 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: This is the half-map A corresponding to the...
| File | emd_17193_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is the half-map A corresponding to the main map from the 3D flex reconstruction job in Cryosparc. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: This is the half-map B corresponding to the...
| File | emd_17193_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is the half-map B corresponding to the main map from the 3D flex reconstruction job in Cryosparc. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: This is the half-map A from the non-uniform...
| File | emd_17193_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is the half-map A from the non-uniform refinement job, the parent job for running 3D flex reconstruction. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: This is the half-map B from the non-uniform...
| File | emd_17193_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is the half-map B from the non-uniform refinement job, the parent job for running 3D flex reconstruction. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of Alanine Serine Cysteine Transporter 2 with Suppressyn
| Entire | Name: Complex of Alanine Serine Cysteine Transporter 2 with Suppressyn |
|---|---|
| Components |
|
-Supramolecule #1: Complex of Alanine Serine Cysteine Transporter 2 with Suppressyn
| Supramolecule | Name: Complex of Alanine Serine Cysteine Transporter 2 with Suppressyn type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Neutral amino acid transporter B(0)
| Macromolecule | Name: Neutral amino acid transporter B(0) / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 57.897273 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MWSHPQFEKV ADPPRDSKGL AAAEPTANGG LALASIEDQG AAAGGYCGSR DQVRRFLRRN LLVLLTVSGV LAGVALGLGV RGAGGGLAL SRAQLTYFAF PGELLLRLLR MIILPLVVCS LIGGAASLDP GALGRLGAWA LLFFLVTTLL ASALGVGLAL A LQPGAASA ...String: MWSHPQFEKV ADPPRDSKGL AAAEPTANGG LALASIEDQG AAAGGYCGSR DQVRRFLRRN LLVLLTVSGV LAGVALGLGV RGAGGGLAL SRAQLTYFAF PGELLLRLLR MIILPLVVCS LIGGAASLDP GALGRLGAWA LLFFLVTTLL ASALGVGLAL A LQPGAASA AITASVGAAG SAENAPSKEV LDSFLDLARN IFPSNLVSAA FRSYSTTYEE RTITGTRVKV PVGQEVEGMN IL GLVVFAI VFGVALRKLG PEGEELIRFF NSFNEATMVL VSWIMWYAPV GIMFLVASKI VEMEDVVLLF TSLGKYIFCC ILG HAIHGL IVLPLIYFAF TRKNPYRFLL GLLTPLATAF GTSSSSATLP LMMKCVEENN GVDKRISRFI LPIGATVNMD GAAI FQCVA AVFIAQLNNV PLNFGQIITI LVTATASSVG AAGIPAGGVL TLAIILEAIG LPTHDLSLIL AVDWLVDRTT TVVNV EGDA LGAGILQHLN DKTMKKSEEQ ELIQVKSELP LDPLPVPTEE GNPLLKHYRG PAGDATVASE KESVM |
-Macromolecule #2: Suppressyn
| Macromolecule | Name: Suppressyn / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 18.152035 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MACIYPTTFY TSLPTKSLNM GISLTTILIL SVAVLLSTAA PPSCRECYQS LHYRGEMQQY FTYHTHIERS CYGNLIEECV ESGKSYYKV KNLGVCGSRN GAICPRGKQW LCFTKIGQWG VNTQVLEDIK REQIIAKAKA SKPTTPPENR PRHFHSFIQK L UniProtKB: Suppressyn |
-Macromolecule #3: ALANINE
| Macromolecule | Name: ALANINE / type: ligand / ID: 3 / Number of copies: 2 / Formula: ALA |
|---|---|
| Molecular weight | Theoretical: 89.093 Da |
| Chemical component information | 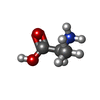 ChemComp-ALA: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.9 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| ||||||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 10 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR | ||||||||||||||||||
| Vitrification | Cryogen name: NITROGEN / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number grids imaged: 1 / Number real images: 8400 / Average electron dose: 42.61 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.8 µm |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)