[English] 日本語
 Yorodumi
Yorodumi- EMDB-17033: Pol I bound to extended and displaced DNA section - open conformation -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Pol I bound to extended and displaced DNA section - open conformation | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | DNA Polymerase I / Okazaki fragment maturation / DNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology information5'-3' exonuclease activity / 3'-5' exonuclease activity / base-excision repair / DNA-templated DNA replication / double-strand break repair / DNA-directed DNA polymerase / DNA-directed DNA polymerase activity / DNA replication / DNA repair / DNA binding ...5'-3' exonuclease activity / 3'-5' exonuclease activity / base-excision repair / DNA-templated DNA replication / double-strand break repair / DNA-directed DNA polymerase / DNA-directed DNA polymerase activity / DNA replication / DNA repair / DNA binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Botto M / Borsellini A / Lamers MH | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: A four-point molecular handover during Okazaki maturation. Authors: Margherita M Botto / Alessandro Borsellini / Meindert H Lamers /    Abstract: DNA replication introduces thousands of RNA primers into the lagging strand that need to be removed for replication to be completed. In Escherichia coli when the replicative DNA polymerase Pol IIIα ...DNA replication introduces thousands of RNA primers into the lagging strand that need to be removed for replication to be completed. In Escherichia coli when the replicative DNA polymerase Pol IIIα terminates at a previously synthesized RNA primer, DNA Pol I takes over and continues DNA synthesis while displacing the downstream RNA primer. The displaced primer is subsequently excised by an endonuclease, followed by the sealing of the nick by a DNA ligase. Yet how the sequential actions of Pol IIIα, Pol I polymerase, Pol I endonuclease and DNA ligase are coordinated is poorly defined. Here we show that each enzymatic activity prepares the DNA substrate for the next activity, creating an efficient four-point molecular handover. The cryogenic-electron microscopy structure of Pol I bound to a DNA substrate with both an upstream and downstream primer reveals how it displaces the primer in a manner analogous to the monomeric helicases. Moreover, we find that in addition to its flap-directed nuclease activity, the endonuclease domain of Pol I also specifically cuts at the RNA-DNA junction, thus marking the end of the RNA primer and creating a 5' end that is a suitable substrate for the ligase activity of LigA once all RNA has been removed. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17033.map.gz emd_17033.map.gz | 4.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17033-v30.xml emd-17033-v30.xml emd-17033.xml emd-17033.xml | 19.6 KB 19.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_17033.png emd_17033.png | 62.8 KB | ||
| Filedesc metadata |  emd-17033.cif.gz emd-17033.cif.gz | 6.6 KB | ||
| Others |  emd_17033_half_map_1.map.gz emd_17033_half_map_1.map.gz emd_17033_half_map_2.map.gz emd_17033_half_map_2.map.gz | 49.6 MB 49.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17033 http://ftp.pdbj.org/pub/emdb/structures/EMD-17033 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17033 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17033 | HTTPS FTP |
-Related structure data
| Related structure data |  8ooyMC  8oo6C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17033.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17033.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.836 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_17033_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
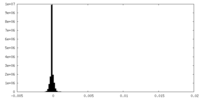
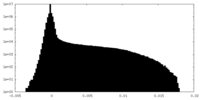
| Density Histograms |
-Half map: #1
| File | emd_17033_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
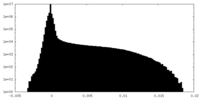
| Density Histograms |
- Sample components
Sample components
-Entire : Escherichia coli DNA Polymerase I in complex with DNA
| Entire | Name: Escherichia coli DNA Polymerase I in complex with DNA |
|---|---|
| Components |
|
-Supramolecule #1: Escherichia coli DNA Polymerase I in complex with DNA
| Supramolecule | Name: Escherichia coli DNA Polymerase I in complex with DNA / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: DNA polymerase I
| Macromolecule | Name: DNA polymerase I / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: DNA-directed DNA polymerase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 67.991461 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GPHDNYVTIL DEETLKAWIA KLEKAPVFAF DTETDSLDNI SANLVGLSFA IEPGVAAYIP VAHDYLDAPD QISRERALEL LKPLLEDEK ALKVGQNLKY DRGILANYGI ELRGIAFDTM LESYILNSVA GRHDMDSLAE RWLKHKTITF EEIAGKGKNQ L TFNQIALE ...String: GPHDNYVTIL DEETLKAWIA KLEKAPVFAF DTETDSLDNI SANLVGLSFA IEPGVAAYIP VAHDYLDAPD QISRERALEL LKPLLEDEK ALKVGQNLKY DRGILANYGI ELRGIAFDTM LESYILNSVA GRHDMDSLAE RWLKHKTITF EEIAGKGKNQ L TFNQIALE EAGRYAAEDA DVTLQLHLKM WPDLQKHKGP LNVFENIEMP LVPVLSRIER NGVKIDPKVL HNHSEELTLR LA ELEKKAH EIAGEEFNLS STKQLQTILF EKQGIKPLKK TPGGAPSTSE EVLEELALDY PLPKVILEYR GLAKLKSTYT DKL PLMINP KTGRVHTSYH QAVTATGRLS STDPNLQNIP VRNEEGRRIR QAFIAPEDYV IVSADYSQIE LRIMAHLSRD KGLL TAFAE GKDIHRATAA EVFGLPLETV TSEQRRSAKA INFGLIYGMS AFGLARQLNI PRKEAQKYMD LYFERYPGVL EYMER TRAQ AKEQGYVETL DGRRLYLPDI KSSNGARRAA AERAAINAPM QGTAADIIKR AMIAVDAWLQ AEQPRVRMIM QVHDEL VFE VHKDDVDAVA KQIHQLMENC TRLDVPLLVE VGSGENWDQA H UniProtKB: DNA polymerase I |
-Macromolecule #2: Template DNA
| Macromolecule | Name: Template DNA / type: other / ID: 2 / Number of copies: 1 Classification: polydeoxyribonucleotide/polyribonucleotide hybrid |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 8.374174 KDa |
| Sequence | String: AACG(DT)CG(DT)GA C(DT)GGGAAAAC CC(DT)GGC |
-Macromolecule #3: Extending Primer
| Macromolecule | Name: Extending Primer / type: other / ID: 3 / Number of copies: 1 Classification: polydeoxyribonucleotide/polyribonucleotide hybrid |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 5.691538 KDa |
| Sequence | String: GCCAGGG(DT)(DT)(DT) (DT)CCCAG(DT)C |
-Macromolecule #4: Displacing Primer
| Macromolecule | Name: Displacing Primer / type: dna / ID: 4 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 2.11341 KDa |
| Sequence | String: (DC)(DG)(DA)(DC)(DG)(DT)(DT) |
-Macromolecule #5: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 5 / Number of copies: 1 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8.5 |
|---|---|
| Sugar embedding | Material: water |
| Vitrification | Cryogen name: ETHANE / Instrument: LEICA PLUNGER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)