[English] 日本語
 Yorodumi
Yorodumi- EMDB-1528: Single copies of Sec61 and TRAP associate with a nontranslating m... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1528 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Single copies of Sec61 and TRAP associate with a nontranslating mammalian ribosome | |||||||||
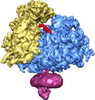
 Map data Map data | This is an average volume from 101000 aligned mammalian ribosome-channel complexes. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | cryo electron microscopy / single particle analysis / ribosome / Sec61 channel / TRAP / ER membrane. co-translational protein translocation | |||||||||
| Function / homology |  Function and homology information Function and homology informationintracellular protein transmembrane transport / SRP-dependent cotranslational protein targeting to membrane, translocation / signal sequence binding / protein transmembrane transporter activity / protein secretion / protein targeting / protein transport / plasma membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 8.7 Å | |||||||||
 Authors Authors | Menetret JF / Hegde RS / Aguiar M / Gygi SP / Park E / Rapoport TA / Akey CW | |||||||||
 Citation Citation |  Journal: Structure / Year: 2008 Journal: Structure / Year: 2008Title: Single copies of Sec61 and TRAP associate with a nontranslating mammalian ribosome. Authors: Jean-François Ménétret / Ramanujan S Hegde / Mike Aguiar / Steven P Gygi / Eunyong Park / Tom A Rapoport / Christopher W Akey /  Abstract: During cotranslational protein translocation, the ribosome associates with a membrane channel, formed by the Sec61 complex, and recruits the translocon-associated protein complex (TRAP). Here we ...During cotranslational protein translocation, the ribosome associates with a membrane channel, formed by the Sec61 complex, and recruits the translocon-associated protein complex (TRAP). Here we report the structure of a ribosome-channel complex from mammalian endoplasmic reticulum in which the channel has been visualized at 11 A resolution. In this complex, single copies of Sec61 and TRAP associate with a nontranslating ribosome and this stoichiometry was verified by quantitative mass spectrometry. A bilayer-like density surrounds the channel and can be attributed to lipid and detergent. The crystal structure of an archaeal homolog of the Sec61 complex was then docked into the map. In this model, two cytoplasmic loops of Sec61 may interact with RNA helices H6, H7, and H50, while the central pore is located below the ribosome tunnel exit. Hence, this copy of Sec61 is positioned to capture and translocate the nascent chain. Finally, we show that mammalian and bacterial ribosome-channel complexes have similar architectures. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1528.map.gz emd_1528.map.gz | 4.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1528-v30.xml emd-1528-v30.xml emd-1528.xml emd-1528.xml | 10.6 KB 10.6 KB | Display Display |  EMDB header EMDB header |
| Images |  EMD1528_Ribi_channel_complex.jpg EMD1528_Ribi_channel_complex.jpg | 177.5 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1528 http://ftp.pdbj.org/pub/emdb/structures/EMD-1528 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1528 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1528 | HTTPS FTP |
-Validation report
| Summary document |  emd_1528_validation.pdf.gz emd_1528_validation.pdf.gz | 331.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_1528_full_validation.pdf.gz emd_1528_full_validation.pdf.gz | 331.4 KB | Display | |
| Data in XML |  emd_1528_validation.xml.gz emd_1528_validation.xml.gz | 5.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1528 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1528 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1528 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1528 | HTTPS FTP |
-Related structure data
| Related structure data |  3dknMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_1528.map.gz / Format: CCP4 / Size: 17.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1528.map.gz / Format: CCP4 / Size: 17.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | This is an average volume from 101000 aligned mammalian ribosome-channel complexes. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.73 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Mammalian native ribosome-channel complex
| Entire | Name: Mammalian native ribosome-channel complex |
|---|---|
| Components |
|
-Supramolecule #1000: Mammalian native ribosome-channel complex
| Supramolecule | Name: Mammalian native ribosome-channel complex / type: sample / ID: 1000 / Number unique components: 3 |
|---|---|
| Molecular weight | Theoretical: 3.77 MDa / Method: from primary sequence |
-Supramolecule #1: ribosome channel complex
| Supramolecule | Name: ribosome channel complex / type: complex / ID: 1 / Name.synonym: ribosome-channel complex Details: Sample solubilized from ER membranes with digitonin. Recombinant expression: No / Ribosome-details: ribosome-eukaryote: ALL |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 3.77 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Details: 30mM Hepes 50mM KAc, 10mM Mg acetate and 1.5% digitonin. |
|---|---|
| Grid | Details: 400 mesh Cu grids with thin continuous carbon film |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 111 K / Instrument: HOMEMADE PLUNGER Details: Vitrification instrument: home-made plunger. in cold room Method: 1 second blot |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Temperature | Average: 93 K |
| Alignment procedure | Legacy - Astigmatism: corrected on-axis at 150K mag |
| Details | data were collected on Oxford and Gatan cryo-holders |
| Date | Jul 27, 2001 |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: OTHER / Digitization - Sampling interval: 4.54 µm / Number real images: 500 / Average electron dose: 15 e/Å2 / Details: Creoscitex Eversmart was used to scan negatives. / Od range: 1 / Bits/pixel: 8 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 51000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 50000 |
| Sample stage | Specimen holder: single tilt / Specimen holder model: GATAN LIQUID NITROGEN |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | 101000 particles were selected using boxer (of EMAN) used as semi-automatic selection program |
|---|---|
| CTF correction | Details: per micrograph |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 8.7 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: EMAN / Number images used: 79000 |
-Atomic model buiding 1
| Initial model | (PDB ID:  2zkr , ) |
|---|---|
| Software | Name: Chimera (UCSF) |
| Details | Protocol: rigid body and manual fitting. The PDBs were fitted using Chimera. The ribosome binding loops of 1RHZ were then flexibly fitted into the riboeome and the stereochemistry was regularized with Coot. |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
| Output model |  PDB-3dkn: |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)