+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-12738 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | 2.12 A cryo-EM structure of Mycobacterium tuberculosis Ferritin | |||||||||
 マップデータ マップデータ | Sharpened map with Locspiral. | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Iron storage / Ferroxidase / Bacterial Ferritin / Octahedral symmetry. / METAL TRANSPORT | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報Mtb iron assimilation by chelation / response to nitrosative stress / encapsulin nanocompartment / iron ion sequestering activity / ferroxidase / intracellular sequestering of iron ion / ferroxidase activity / peptidoglycan-based cell wall / ferric iron binding / ferrous iron binding ...Mtb iron assimilation by chelation / response to nitrosative stress / encapsulin nanocompartment / iron ion sequestering activity / ferroxidase / intracellular sequestering of iron ion / ferroxidase activity / peptidoglycan-based cell wall / ferric iron binding / ferrous iron binding / iron ion transport / response to hypoxia / extracellular region / plasma membrane / cytoplasm / cytosol 類似検索 - 分子機能 | |||||||||
| 生物種 |  Mycobacterium tuberculosis H37Rv (結核菌) Mycobacterium tuberculosis H37Rv (結核菌) | |||||||||
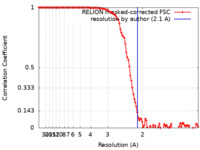
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.1 Å | |||||||||
 データ登録者 データ登録者 | Gijsbers A / Zhang Y | |||||||||
| 資金援助 | European Union,  オランダ, 2件 オランダ, 2件
| |||||||||
 引用 引用 |  ジャーナル: Acta Crystallogr D Struct Biol / 年: 2021 ジャーナル: Acta Crystallogr D Struct Biol / 年: 2021タイトル: Mycobacterium tuberculosis ferritin: a suitable workhorse protein for cryo-EM development. 著者: Abril Gijsbers / Yue Zhang / Ye Gao / Peter J Peters / Raimond B G Ravelli /  要旨: The use of cryo-EM continues to expand worldwide and calls for good-quality standard proteins with simple protocols for their production. Here, a straightforward expression and purification protocol ...The use of cryo-EM continues to expand worldwide and calls for good-quality standard proteins with simple protocols for their production. Here, a straightforward expression and purification protocol is presented that provides an apoferritin, bacterioferritin B (BfrB), from Mycobacterium tuberculosis with high yield and purity. A 2.12 Å resolution cryo-EM structure of BfrB is reported, showing the typical cage-like oligomer constituting of 24 monomers related by 432 symmetry. However, it also contains a unique C-terminal extension (164-181), which loops into the cage region of the shell and provides extra stability to the protein. Part of this region was ambiguous in previous crystal structures but could be built within the cryo-EM map. These findings and this protocol could serve the growing cryo-EM community in characterizing and pushing the limits of their electron microscopes and workflows. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_12738.map.gz emd_12738.map.gz | 22.7 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-12738-v30.xml emd-12738-v30.xml emd-12738.xml emd-12738.xml | 22.3 KB 22.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_12738_fsc.xml emd_12738_fsc.xml | 11.3 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_12738.png emd_12738.png | 70 KB | ||
| マスクデータ |  emd_12738_msk_1.map emd_12738_msk_1.map | 125 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-12738.cif.gz emd-12738.cif.gz | 6.4 KB | ||
| その他 |  emd_12738_additional_1.map.gz emd_12738_additional_1.map.gz emd_12738_half_map_1.map.gz emd_12738_half_map_1.map.gz emd_12738_half_map_2.map.gz emd_12738_half_map_2.map.gz | 20.6 MB 94.1 MB 94.1 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12738 http://ftp.pdbj.org/pub/emdb/structures/EMD-12738 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12738 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12738 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_12738_validation.pdf.gz emd_12738_validation.pdf.gz | 967.2 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_12738_full_validation.pdf.gz emd_12738_full_validation.pdf.gz | 966.8 KB | 表示 | |
| XML形式データ |  emd_12738_validation.xml.gz emd_12738_validation.xml.gz | 19.5 KB | 表示 | |
| CIF形式データ |  emd_12738_validation.cif.gz emd_12738_validation.cif.gz | 24.8 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12738 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12738 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12738 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12738 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_12738.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_12738.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Sharpened map with Locspiral. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.6514 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-マスク #1
| ファイル |  emd_12738_msk_1.map emd_12738_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: Relion postprocessing sharpened map
| ファイル | emd_12738_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Relion postprocessing sharpened map | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_12738_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_12738_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Mycobacterium tuberculosis ferritin
| 全体 | 名称: Mycobacterium tuberculosis ferritin |
|---|---|
| 要素 |
|
-超分子 #1: Mycobacterium tuberculosis ferritin
| 超分子 | 名称: Mycobacterium tuberculosis ferritin / タイプ: organelle_or_cellular_component / ID: 1 / 親要素: 0 / 含まれる分子: #1 / 詳細: Mycobacterium tuberculosis ferritin |
|---|---|
| 由来(天然) | 生物種:  Mycobacterium tuberculosis H37Rv (結核菌) / 株: H37Rv Mycobacterium tuberculosis H37Rv (結核菌) / 株: H37Rv |
| 分子量 | 理論値: 490 KDa |
-分子 #1: Ferritin BfrB
| 分子 | 名称: Ferritin BfrB / タイプ: protein_or_peptide / ID: 1 / コピー数: 24 / 光学異性体: LEVO / EC番号: ferroxidase |
|---|---|
| 由来(天然) | 生物種:  Mycobacterium tuberculosis H37Rv (結核菌) Mycobacterium tuberculosis H37Rv (結核菌) |
| 分子量 | 理論値: 20.463936 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MTEYEGPKTK FHALMQEQIH NEFTAAQQYV AIAVYFDSED LPQLAKHFYS QAVEERNHAM MLVQHLLDRD LRVEIPGVDT VRNQFDRPR EALALALDQE RTVTDQVGRL TAVARDEGDF LGEQFMQWFL QEQIEEVALM ATLVRVADRA GANLFELENF V AREVDVAP AASGAPHAAG GRL UniProtKB: Bacterioferritin BfrB |
-分子 #2: water
| 分子 | 名称: water / タイプ: ligand / ID: 2 / コピー数: 1392 / 式: HOH |
|---|---|
| 分子量 | 理論値: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 11 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 8 構成要素:
| |||||||||
| グリッド | モデル: UltrAuFoil / 材質: GOLD / メッシュ: 300 / 支持フィルム - 材質: GOLD / 支持フィルム - トポロジー: HOLEY ARRAY / 支持フィルム - Film thickness: 2000 / 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 時間: 40 sec. / 前処理 - 雰囲気: AIR / 前処理 - 気圧: 0.01 kPa | |||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277 K / 装置: FEI VITROBOT MARK IV | |||||||||
| 詳細 | This sample was monodisperse. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 温度 | 最低: 93.0 K / 最高: 105.0 K |
| 特殊光学系 | エネルギーフィルター - 名称: GIF Bioquantum / エネルギーフィルター - スリット幅: 20 eV |
| 詳細 | Basic direct alignments were done as well as astigmatism and coma alignment using AutoCTF |
| 撮影 | フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) デジタル化 - サイズ - 横: 5760 pixel / デジタル化 - サイズ - 縦: 4092 pixel / 撮影したグリッド数: 1 / 実像数: 2518 / 平均露光時間: 1.3 sec. / 平均電子線量: 40.0 e/Å2 詳細: Images were acquired in superresolution counting mode at a speed of 64 movies/hour and stored as tiff lzw non-gain normalised |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 50.0 µm / 最大 デフォーカス(補正後): 2.0 µm / 最小 デフォーカス(補正後): 0.2 µm / 倍率(補正後): 76757 / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm 最大 デフォーカス(公称値): 1.4000000000000001 µm 最小 デフォーカス(公称値): 0.4 µm / 倍率(公称値): 130000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 初期モデル | PDB ID: Chain - Chain ID: A / Chain - Residue range: 10-181 / Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| 精密化 | 空間: REAL / プロトコル: OTHER / 温度因子: 28.1 / 当てはまり具合の基準: Correlation coefficient |
| 得られたモデル |  PDB-7o6e: |
 ムービー
ムービー コントローラー
コントローラー


















 Z
Z Y
Y X
X