[English] 日本語
 Yorodumi
Yorodumi- EMDB-12631: Mammalian pre-termination 80S ribosome with Hybrid P/E- and A/P-s... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12631 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Mammalian pre-termination 80S ribosome with Hybrid P/E- and A/P-site tRNA's bound by Blasticidin S. | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Inhibitor 80S Termination Complex Blasticidin S Translation NMD / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationL13a-mediated translational silencing of Ceruloplasmin expression / SRP-dependent cotranslational protein targeting to membrane / Major pathway of rRNA processing in the nucleolus and cytosol / Formation of a pool of free 40S subunits / GTP hydrolysis and joining of the 60S ribosomal subunit / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Translation initiation complex formation / Formation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition ...L13a-mediated translational silencing of Ceruloplasmin expression / SRP-dependent cotranslational protein targeting to membrane / Major pathway of rRNA processing in the nucleolus and cytosol / Formation of a pool of free 40S subunits / GTP hydrolysis and joining of the 60S ribosomal subunit / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Translation initiation complex formation / Formation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition / Major pathway of rRNA processing in the nucleolus and cytosol / GTP hydrolysis and joining of the 60S ribosomal subunit / L13a-mediated translational silencing of Ceruloplasmin expression / SRP-dependent cotranslational protein targeting to membrane / Formation of a pool of free 40S subunits / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / laminin receptor activity / ubiquitin ligase inhibitor activity / 90S preribosome / endonucleolytic cleavage to generate mature 3'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / positive regulation of signal transduction by p53 class mediator / phagocytic cup / endonucleolytic cleavage in ITS1 to separate SSU-rRNA from 5.8S rRNA and LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / translation regulator activity / rough endoplasmic reticulum / ribosomal small subunit export from nucleus / laminin binding / gastrulation / MDM2/MDM4 family protein binding / cytosolic ribosome / class I DNA-(apurinic or apyrimidinic site) endonuclease activity / DNA-(apurinic or apyrimidinic site) lyase / ribosomal large subunit biogenesis / maturation of LSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / positive regulation of apoptotic signaling pathway / maturation of SSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / maturation of SSU-rRNA / small-subunit processome / spindle / cytoplasmic ribonucleoprotein granule / rRNA processing / positive regulation of canonical Wnt signaling pathway / rhythmic process / regulation of translation / antimicrobial humoral immune response mediated by antimicrobial peptide / large ribosomal subunit / ribosomal small subunit assembly / virus receptor activity / ribosome binding / ribosomal small subunit biogenesis / 5S rRNA binding / small ribosomal subunit / ribosomal large subunit assembly / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / large ribosomal subunit rRNA binding / killing of cells of another organism / cytosolic large ribosomal subunit / defense response to Gram-negative bacterium / perikaryon / cytoplasmic translation / cell differentiation / tRNA binding / mitochondrial inner membrane / postsynaptic density / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / cell division / DNA repair / mRNA binding / apoptotic process / centrosome / synapse / dendrite / nucleolus / perinuclear region of cytoplasm / endoplasmic reticulum / Golgi apparatus / DNA binding / RNA binding / zinc ion binding / membrane / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Powers KT / Yadav SKN | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2021 Journal: Nucleic Acids Res / Year: 2021Title: Blasticidin S inhibits mammalian translation and enhances production of protein encoded by nonsense mRNA. Authors: Kyle T Powers / Flint Stevenson-Jones / Sathish K N Yadav / Beate Amthor / Joshua C Bufton / Ufuk Borucu / Dakang Shen / Jonas P Becker / Daria Lavysh / Matthias W Hentze / Andreas E Kulozik ...Authors: Kyle T Powers / Flint Stevenson-Jones / Sathish K N Yadav / Beate Amthor / Joshua C Bufton / Ufuk Borucu / Dakang Shen / Jonas P Becker / Daria Lavysh / Matthias W Hentze / Andreas E Kulozik / Gabriele Neu-Yilik / Christiane Schaffitzel /   Abstract: Deciphering translation is of paramount importance for the understanding of many diseases, and antibiotics played a pivotal role in this endeavour. Blasticidin S (BlaS) targets translation by binding ...Deciphering translation is of paramount importance for the understanding of many diseases, and antibiotics played a pivotal role in this endeavour. Blasticidin S (BlaS) targets translation by binding to the peptidyl transferase center of the large ribosomal subunit. Using biochemical, structural and cellular approaches, we show here that BlaS inhibits both translation elongation and termination in Mammalia. Bound to mammalian terminating ribosomes, BlaS distorts the 3'CCA tail of the P-site tRNA to a larger extent than previously reported for bacterial ribosomes, thus delaying both, peptide bond formation and peptidyl-tRNA hydrolysis. While BlaS does not inhibit stop codon recognition by the eukaryotic release factor 1 (eRF1), it interferes with eRF1's accommodation into the peptidyl transferase center and subsequent peptide release. In human cells, BlaS inhibits nonsense-mediated mRNA decay and, at subinhibitory concentrations, modulates translation dynamics at premature termination codons leading to enhanced protein production. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12631.map.gz emd_12631.map.gz | 16 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12631-v30.xml emd-12631-v30.xml emd-12631.xml emd-12631.xml | 105.5 KB 105.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12631_fsc.xml emd_12631_fsc.xml | 10.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_12631.png emd_12631.png | 100.5 KB | ||
| Masks |  emd_12631_msk_1.map emd_12631_msk_1.map | 103 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-12631.cif.gz emd-12631.cif.gz | 20.6 KB | ||
| Others |  emd_12631_half_map_1.map.gz emd_12631_half_map_1.map.gz emd_12631_half_map_2.map.gz emd_12631_half_map_2.map.gz | 80.6 MB 80.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12631 http://ftp.pdbj.org/pub/emdb/structures/EMD-12631 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12631 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12631 | HTTPS FTP |
-Related structure data
| Related structure data |  7nwgMC  7nwhC  7nwiC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12631.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12631.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12631_msk_1.map emd_12631_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_12631_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_12631_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Mammalian pre-termination 80S ribosome with Hybrid-P/E and -A/P s...
+Supramolecule #1: Mammalian pre-termination 80S ribosome with Hybrid-P/E and -A/P s...
+Macromolecule #1: 28S Ribosomal RNA
+Macromolecule #2: 5S Ribosomal RNA
+Macromolecule #3: 5.8S Ribosomal RNA
+Macromolecule #4: 18S Ribosomal RNA
+Macromolecule #79: A/P-Site tRNA
+Macromolecule #82: mRNA
+Macromolecule #83: P/E-Site tRNA
+Macromolecule #5: 40S ribosomal protein SA
+Macromolecule #6: 40S ribosomal protein S3a
+Macromolecule #7: 40S ribosomal protein S2
+Macromolecule #8: 40S ribosomal protein S3
+Macromolecule #9: S4
+Macromolecule #10: Ribosomal protein S5
+Macromolecule #11: 40S ribosomal protein S6
+Macromolecule #12: ribosomal protein eS7
+Macromolecule #13: 40S ribosomal protein S8
+Macromolecule #14: 40S ribosomal protein S9
+Macromolecule #15: 40S ribosomal protein S10
+Macromolecule #16: 40S ribosomal protein S11
+Macromolecule #17: 40S ribosomal protein S12
+Macromolecule #18: ribosomal protein uS15
+Macromolecule #19: uS11
+Macromolecule #20: 40S ribosomal protein uS19
+Macromolecule #21: Ribosomal protein S16
+Macromolecule #22: 40S ribosomal protein S17
+Macromolecule #23: 40S ribosomal protein uS13
+Macromolecule #24: S19
+Macromolecule #25: 40S ribosomal protein S20
+Macromolecule #26: 40S ribosomal protein S21
+Macromolecule #27: Ribosomal protein S15a
+Macromolecule #28: Ribosomal protein S23
+Macromolecule #29: 40S ribosomal protein S24
+Macromolecule #30: ribosomal protein eS25
+Macromolecule #31: 40S ribosomal protein S26
+Macromolecule #32: 40S ribosomal protein S27
+Macromolecule #33: 40S ribosomal protein S28
+Macromolecule #34: uS14
+Macromolecule #35: 40S ribosomal protein S30
+Macromolecule #36: eS31
+Macromolecule #37: ribosomal protein RACK1
+Macromolecule #38: L8
+Macromolecule #39: uL3
+Macromolecule #40: uL4
+Macromolecule #41: 60S ribosomal protein L5
+Macromolecule #42: 60S ribosomal protein L6
+Macromolecule #43: uL30
+Macromolecule #44: L7a
+Macromolecule #45: L9
+Macromolecule #46: 60S ribosomal protein L10
+Macromolecule #47: 60S ribosomal protein L11
+Macromolecule #48: L13
+Macromolecule #49: Ribosomal protein L14
+Macromolecule #50: Ribosomal protein L15
+Macromolecule #51: 60S RIBOSOMAL PROTEIN UL13
+Macromolecule #52: uL22
+Macromolecule #53: L18
+Macromolecule #54: 60S ribosomal protein L19
+Macromolecule #55: 60S ribosomal protein L18a
+Macromolecule #56: eL21
+Macromolecule #57: L22
+Macromolecule #58: eL14
+Macromolecule #59: uL23
+Macromolecule #60: Ribosomal protein L26
+Macromolecule #61: 60S ribosomal protein L27
+Macromolecule #62: uL15
+Macromolecule #63: eL29
+Macromolecule #64: eL30
+Macromolecule #65: eL31
+Macromolecule #66: eL32
+Macromolecule #67: eL33
+Macromolecule #68: 60S ribosomal protein L34
+Macromolecule #69: uL29
+Macromolecule #70: 60S ribosomal protein L36
+Macromolecule #71: Ribosomal protein L37
+Macromolecule #72: L38
+Macromolecule #73: eL39
+Macromolecule #74: 60S RIBOSOMAL PROTEIN EL40
+Macromolecule #75: 60s ribosomal protein l41
+Macromolecule #76: eL42
+Macromolecule #77: ribosomal protein eL43
+Macromolecule #78: eL28
+Macromolecule #80: 60S acidic ribosomal protein P0
+Macromolecule #81: uL12
+Macromolecule #84: Ribosomal protein
+Macromolecule #85: Sec61Beta
+Macromolecule #86: 60S ribosomal protein L24-like protein
+Macromolecule #87: MAGNESIUM ION
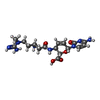
+Macromolecule #88: BLASTICIDIN S
+Macromolecule #89: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.63 mg/mL |
|---|---|
| Buffer | pH: 7.4 Details: 50 mM HEPES pH 7.4 100 mM KOAc 5 mM Mg(Oac)2 1mM DTT |
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 70 % / Chamber temperature: 288.15 K / Instrument: LEICA EM GP Details: 30s sample incubation on grid followed by 1.1s blotting time.. |
| Details | 165nM (OD260nm~10.5) |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI ARCTICA |
|---|---|
| Details | Collected in super-resolution mode (0.675A pixel size) |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Number grids imaged: 1 / Number real images: 3500 / Average exposure time: 8.0 sec. / Average electron dose: 41.9 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 2.0 µm / Calibrated defocus min: 0.4 µm / Calibrated magnification: 79000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)