+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12297 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Ovine rBAT ectodomain homodimer, C2-symmetry | |||||||||
 Map data Map data | Postprocessed map | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationglucan 1,4-alpha-maltotriohydrolase activity / oligo-1,6-glucosidase activity / maltose catabolic process / : / sucrose alpha-glucosidase activity / sucrose catabolic process / alpha-amylase activity / amino acid transport Similarity search - Function | |||||||||
| Biological species |  | |||||||||
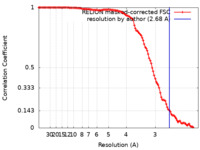
| Method | single particle reconstruction / cryo EM / Resolution: 2.68 Å | |||||||||
 Authors Authors | Lee Y / Kuehlbrandt W | |||||||||
| Funding support |  Germany, 2 items Germany, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Ca-mediated higher-order assembly of heterodimers in amino acid transport system b biogenesis and cystinuria. Authors: Yongchan Lee / Pattama Wiriyasermkul / Pornparn Kongpracha / Satomi Moriyama / Deryck J Mills / Werner Kühlbrandt / Shushi Nagamori /   Abstract: Cystinuria is a genetic disorder characterized by overexcretion of dibasic amino acids and cystine, causing recurrent kidney stones and kidney failure. Mutations of the regulatory glycoprotein rBAT ...Cystinuria is a genetic disorder characterized by overexcretion of dibasic amino acids and cystine, causing recurrent kidney stones and kidney failure. Mutations of the regulatory glycoprotein rBAT and the amino acid transporter bAT, which constitute system b, are linked to type I and non-type I cystinuria respectively and they exhibit distinct phenotypes due to protein trafficking defects or catalytic inactivation. Here, using electron cryo-microscopy and biochemistry, we discover that Ca mediates higher-order assembly of system b. Ca stabilizes the interface between two rBAT molecules, leading to super-dimerization of bAT-rBAT, which in turn facilitates N-glycan maturation and protein trafficking. A cystinuria mutant T216M and mutations of the Ca site of rBAT cause the loss of higher-order assemblies, resulting in protein trapping at the ER and the loss of function. These results provide the molecular basis of system b biogenesis and type I cystinuria and serve as a guide to develop new therapeutic strategies against it. More broadly, our findings reveal an unprecedented link between transporter oligomeric assembly and protein-trafficking diseases. #1:  Journal: Biorxiv / Year: 2021 Journal: Biorxiv / Year: 2021Title: Ca 2+ -mediated higher-order assembly of b 0,+ AT-rBAT is a key step for system b 0,+ biogenesis and cystinuria Authors: Lee Y / Wiriyasermkul P / Moriyama S / Mills DJ / Kuhlbrandt W / Nagamori S | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12297.map.gz emd_12297.map.gz | 9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12297-v30.xml emd-12297-v30.xml emd-12297.xml emd-12297.xml | 15.9 KB 15.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12297_fsc.xml emd_12297_fsc.xml | 11.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_12297.png emd_12297.png | 91.1 KB | ||
| Masks |  emd_12297_msk_1.map emd_12297_msk_1.map | 125 MB |  Mask map Mask map | |
| Others |  emd_12297_half_map_1.map.gz emd_12297_half_map_1.map.gz emd_12297_half_map_2.map.gz emd_12297_half_map_2.map.gz | 96.7 MB 96.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12297 http://ftp.pdbj.org/pub/emdb/structures/EMD-12297 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12297 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12297 | HTTPS FTP |
-Related structure data
| Related structure data |  7nf7MC  7nf6C  7nf8C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12297.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12297.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Postprocessed map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09856 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
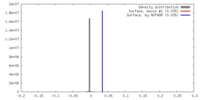
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12297_msk_1.map emd_12297_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
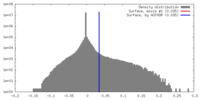
| Density Histograms |
-Half map: Half map 1
| File | emd_12297_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
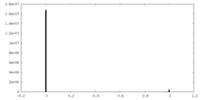
| Density Histograms |
-Half map: Half map 2
| File | emd_12297_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
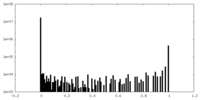
| Density Histograms |
- Sample components
Sample components
-Entire : Ovine rBAT ectodomain homodimer
| Entire | Name: Ovine rBAT ectodomain homodimer |
|---|---|
| Components |
|
-Supramolecule #1: Ovine rBAT ectodomain homodimer
| Supramolecule | Name: Ovine rBAT ectodomain homodimer / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 79 kDa/nm |
-Macromolecule #1: neutral and basic amino acid transport protein rBAT
| Macromolecule | Name: neutral and basic amino acid transport protein rBAT / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 78.754281 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: GSAEDKSKRD SIGLNAKEGQ TNNGFVQNED ILETDLDPSS PAAGPQHNTV DILGPGEPDV KDVRPYAGMP KEVLFQFSGQ ARYRIPREV LFWLTVASVL LLIAATIAII AISPKCLDWW QAGPMYQIYP RSFRDSNKDG DGDLKGIQDK LDYITTLNIK T VWITSFYK ...String: GSAEDKSKRD SIGLNAKEGQ TNNGFVQNED ILETDLDPSS PAAGPQHNTV DILGPGEPDV KDVRPYAGMP KEVLFQFSGQ ARYRIPREV LFWLTVASVL LLIAATIAII AISPKCLDWW QAGPMYQIYP RSFRDSNKDG DGDLKGIQDK LDYITTLNIK T VWITSFYK SSLKDFRHAV EDFQEIDPIF GTMKDFENLV AAIHDKGLKL IIDFIPNHTS DKHAWFQWSR NRTGKYTDYY IW HDCNYEN GTTIPPNNWL SVYGNSSWHF DEVRKQCYFH QFMKEQPDLN FRNPDVQEEI KEIIQFWLSK GVDGFSFNAL QYL LEAKHL RDEAQVNKTQ IPDTVTHYSQ LHHDFTTTQV GMHDIVRSFR QTMNQYSREP GRYRFMGTEA HGESITETMV YYGL PFIQE ADFPFNSYLS KLDKPSGNSV SEVITSWLEN MPEGKWPNWM TGGPDNVRLT SRLGEKYVNI MNMLVFTLPG TPITY YGEE IGMRNILAAN LNENYDTGTL FSKSPMQWDN SSNAGFSEGN HTWLPTSSDY HTVNVDVQKT QPRSALKLYQ ELSLLH ANE LLLSRGWFCY LRNDNHSIMY TRELDGINKV FLMVLNFGES SLLNLKEMIS NIPTRVRIRL STSSAYSGRE VDTHAVT LA SGEGLILEYN TGNLLHRQTA FKDRCFVSNR ACYSRVLNIL YSLC |
-Macromolecule #3: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 3 / Number of copies: 1 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Macromolecule #4: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 4 / Number of copies: 4 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)