+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-0598 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structure of a drug-like molecule stalled ribosome nascent chain complex (PCSK9_RNC) in non-rotated state with PP tRNA | |||||||||
 マップデータ マップデータ | PF846 stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA. Map is sharpened. | |||||||||
 試料 試料 |
| |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報low-density lipoprotein particle receptor catabolic process / negative regulation of receptor-mediated endocytosis involved in cholesterol transport / extrinsic component of external side of plasma membrane / negative regulation of sodium ion import across plasma membrane / PCSK9-LDLR complex / PCSK9-AnxA2 complex / negative regulation of receptor recycling / apolipoprotein receptor binding / very-low-density lipoprotein particle binding / low-density lipoprotein particle binding ...low-density lipoprotein particle receptor catabolic process / negative regulation of receptor-mediated endocytosis involved in cholesterol transport / extrinsic component of external side of plasma membrane / negative regulation of sodium ion import across plasma membrane / PCSK9-LDLR complex / PCSK9-AnxA2 complex / negative regulation of receptor recycling / apolipoprotein receptor binding / very-low-density lipoprotein particle binding / low-density lipoprotein particle binding / positive regulation of low-density lipoprotein particle receptor catabolic process / LDL clearance / lipoprotein metabolic process / very-low-density lipoprotein particle receptor binding / translation at presynapse / embryonic brain development / exit from mitosis / eukaryotic 80S initiation complex / negative regulation of protein neddylation / negative regulation of receptor internalization / optic nerve development / response to insecticide / COPII-coated ER to Golgi transport vesicle / negative regulation of endoplasmic reticulum unfolded protein response / regulation of translation involved in cellular response to UV / oxidized pyrimidine DNA binding / response to TNF agonist / positive regulation of base-excision repair / axial mesoderm development / negative regulation of formation of translation preinitiation complex / regulation of G1 to G0 transition / ribosomal protein import into nucleus / positive regulation of respiratory burst involved in inflammatory response / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage / endolysosome membrane / positive regulation of gastrulation / 90S preribosome assembly / protein tyrosine kinase inhibitor activity / protein-DNA complex disassembly / sodium channel inhibitor activity / IRE1-RACK1-PP2A complex / positive regulation of endodeoxyribonuclease activity / nucleolus organization / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / positive regulation of Golgi to plasma membrane protein transport / retinal ganglion cell axon guidance / TNFR1-mediated ceramide production / negative regulation of low-density lipoprotein particle clearance / negative regulation of DNA repair / negative regulation of RNA splicing / signaling receptor inhibitor activity / GAIT complex / positive regulation of DNA damage response, signal transduction by p53 class mediator / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / supercoiled DNA binding / TORC2 complex binding / alpha-beta T cell differentiation / neural crest cell differentiation / G1 to G0 transition / NF-kappaB complex / positive regulation of ubiquitin-protein transferase activity / cysteine-type endopeptidase activator activity involved in apoptotic process / oxidized purine DNA binding / negative regulation of intrinsic apoptotic signaling pathway in response to hydrogen peroxide / ubiquitin-like protein conjugating enzyme binding / negative regulation of bicellular tight junction assembly / regulation of establishment of cell polarity / lysosomal transport / middle ear morphogenesis / negative regulation of phagocytosis / rRNA modification in the nucleus and cytosol / triglyceride metabolic process / Formation of the ternary complex, and subsequently, the 43S complex / erythrocyte homeostasis / low-density lipoprotein particle receptor binding / cytoplasmic side of rough endoplasmic reticulum membrane / laminin receptor activity / negative regulation of ubiquitin protein ligase activity / protein kinase A binding / ion channel inhibitor activity / pigmentation / Ribosomal scanning and start codon recognition / homeostatic process / Translation initiation complex formation / positive regulation of mitochondrial depolarization / macrophage chemotaxis / positive regulation of T cell receptor signaling pathway / fibroblast growth factor binding / negative regulation of Wnt signaling pathway / lung morphogenesis / monocyte chemotaxis / positive regulation of activated T cell proliferation / positive regulation of natural killer cell proliferation / negative regulation of translational frameshifting / Protein hydroxylation / TOR signaling / BH3 domain binding / SARS-CoV-1 modulates host translation machinery / regulation of cell division / mTORC1-mediated signalling 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.9 Å | |||||||||
 データ登録者 データ登録者 | Li W / Cate JHD | |||||||||
| 資金援助 |  米国, 1件 米国, 1件
| |||||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2019 ジャーナル: Nat Struct Mol Biol / 年: 2019タイトル: Structural basis for selective stalling of human ribosome nascent chain complexes by a drug-like molecule. 著者: Wenfei Li / Fred R Ward / Kim F McClure / Stacey Tsai-Lan Chang / Elizabeth Montabana / Spiros Liras / Robert G Dullea / Jamie H D Cate /  要旨: The drug-like molecule PF-06446846 (PF846) binds the human ribosome and selectively blocks the translation of a small number of proteins by an unknown mechanism. In structures of PF846-stalled human ...The drug-like molecule PF-06446846 (PF846) binds the human ribosome and selectively blocks the translation of a small number of proteins by an unknown mechanism. In structures of PF846-stalled human ribosome nascent chain complexes, PF846 binds in the ribosome exit tunnel in a eukaryotic-specific pocket formed by 28S ribosomal RNA, and alters the path of the nascent polypeptide chain. PF846 arrests the translating ribosome in the rotated state of translocation, in which the peptidyl-transfer RNA 3'-CCA end is improperly docked in the peptidyl transferase center. Selections of messenger RNAs from mRNA libraries using translation extracts reveal that PF846 can stall translation elongation, arrest termination or even enhance translation, depending on nascent chain sequence context. These results illuminate how a small molecule selectively targets translation by the human ribosome, and provides a foundation for developing small molecules that modulate the production of proteins of therapeutic interest. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_0598.map.gz emd_0598.map.gz | 229 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-0598-v30.xml emd-0598-v30.xml emd-0598.xml emd-0598.xml | 17.2 KB 17.2 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_0598_fsc.xml emd_0598_fsc.xml | 14.2 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_0598.png emd_0598.png | 163.5 KB | ||
| その他 |  emd_0598_additional.map.gz emd_0598_additional.map.gz emd_0598_half_map_1.map.gz emd_0598_half_map_1.map.gz emd_0598_half_map_2.map.gz emd_0598_half_map_2.map.gz | 193.1 MB 193.8 MB 193.8 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0598 http://ftp.pdbj.org/pub/emdb/structures/EMD-0598 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0598 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0598 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_0598_validation.pdf.gz emd_0598_validation.pdf.gz | 714.2 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_0598_full_validation.pdf.gz emd_0598_full_validation.pdf.gz | 713.8 KB | 表示 | |
| XML形式データ |  emd_0598_validation.xml.gz emd_0598_validation.xml.gz | 19.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0598 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0598 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0598 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0598 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  6olzMC  0526C  0596C  0597C  0599C  0600C  0601C  6oleC  6olfC  6olgC  6oliC  6om0C  6om7C C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_0598.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_0598.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | PF846 stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA. Map is sharpened. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.22 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
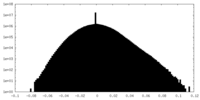
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-追加マップ: PF846 stalled human ribosome nascent chain complex (PCSK9-RNC)...
| ファイル | emd_0598_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | PF846 stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA. Map is not sharpened. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
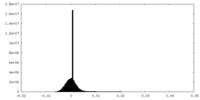
| 密度ヒストグラム |
-ハーフマップ: Half map of PF846 stalled human ribosome nascent...
| ファイル | emd_0598_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map of PF846 stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
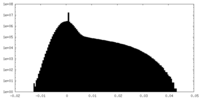
| 密度ヒストグラム |
-ハーフマップ: Half map of PF846 stalled human ribosome nascent...
| ファイル | emd_0598_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map of PF846 stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
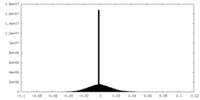
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Drug-like molecule stalled human ribosome nascent chain complex (...
| 全体 | 名称: Drug-like molecule stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA |
|---|---|
| 要素 |
|
-超分子 #1: Drug-like molecule stalled human ribosome nascent chain complex (...
| 超分子 | 名称: Drug-like molecule stalled human ribosome nascent chain complex (PCSK9-RNC) in non-rotated state with PP tRNA タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 組換発現 | 生物種:  Homo sapiens (ヒト) / 組換細胞: HeLa Homo sapiens (ヒト) / 組換細胞: HeLa |
| 分子量 | 理論値: 4.3 MDa |
-分子 #1: human ribosome with nascent chain
| 分子 | 名称: human ribosome with nascent chain / タイプ: rna / ID: 1 詳細: Nascent chain sequence including CDH1 domain V and PCSK9 1-35 |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: SDVNDNAPIP EPRTIFFCER NPKPQVINII DADLPPNTSP FTAELTHGAS ANWTIQYNDP TQESIILKPK MALEVGDYKI NLKLMDNQNK DQVTTLEVSV CDCEGAAGVC RKAQPVEAGL QIPAILGILG MGTVSSRRS WWPLPLLLLL LLLLGPAGAR AQEDED |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.8 |
|---|---|
| グリッド | 詳細: unspecified |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 4 K |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 最終 再構成 | 解像度のタイプ: BY AUTHOR / 解像度: 3.9 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 使用した粒子像数: 7214 |
|---|---|
| 初期 角度割当 | タイプ: ANGULAR RECONSTITUTION |
| 最終 角度割当 | タイプ: ANGULAR RECONSTITUTION |
| FSC曲線 (解像度の算出) | 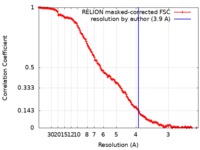 |
-原子モデル構築 1
| 精密化 | 空間: REAL / 温度因子: 80 |
|---|---|
| 得られたモデル |  PDB-6olz: |
 ムービー
ムービー コントローラー
コントローラー
















































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)