[English] 日本語
 Yorodumi
Yorodumi- EMDB-33403: 3.1 Angstrom cryoEM icosahedral reconstruction of mud crab reovirus -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | 3.1 Angstrom cryoEM icosahedral reconstruction of mud crab reovirus | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Reovirus / ds-RNA virus / VIRUS | |||||||||
| Function / homology | VP3 / VP11 / VP12 Function and homology information Function and homology information | |||||||||
| Biological species |  Scylla serrata reovirus SZ-2007 Scylla serrata reovirus SZ-2007 | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Zhang Q / Gao Y | |||||||||
| Funding support |  China, 2 items China, 2 items
| |||||||||
 Citation Citation |  Journal: PLoS Pathog / Year: 2023 Journal: PLoS Pathog / Year: 2023Title: The structure of a 12-segmented dsRNA reovirus: New insights into capsid stabilization and organization. Authors: Qinfen Zhang / Yuanzhu Gao / Matthew L Baker / Shanshan Liu / Xudong Jia / Haidong Xu / Jianguo He / Jason T Kaelber / Shaoping Weng / Wen Jiang /   Abstract: Infecting a wide range of hosts, members of Reovirales (formerly Reoviridae) consist of a genome with different numbers of segmented double stranded RNAs (dsRNA) encapsulated by a proteinaceous shell ...Infecting a wide range of hosts, members of Reovirales (formerly Reoviridae) consist of a genome with different numbers of segmented double stranded RNAs (dsRNA) encapsulated by a proteinaceous shell and carry out genome replication and transcription inside the virion. Several cryo-electron microscopy (cryo-EM) structures of reoviruses with 9, 10 or 11 segmented dsRNA genomes have revealed insights into genome arrangement and transcription. However, the structure and genome arrangement of 12-segmented Reovirales members remain poorly understood. Using cryo-EM, we determined the structure of mud crab reovirus (MCRV), a 12-segmented dsRNA virus that is a putative member of Reovirales in the non-turreted Sedoreoviridae family, to near-atomic resolutions with icosahedral symmetry (3.1 Å) and without imposing icosahedral symmetry (3.4 Å). These structures revealed the organization of the major capsid proteins in two layers: an outer T = 13 layer consisting of VP12 trimers and unique VP11 clamps, and an inner T = 1 layer consisting of VP3 dimers. Additionally, ten RNA dependent RNA polymerases (RdRp) were well resolved just below the VP3 layer but were offset from the 5-fold axes and arranged with D5 symmetry, which has not previously been seen in other members of Reovirales. The N-termini of VP3 were shown to adopt four unique conformations; two of which anchor the RdRps, while the other two conformations are likely involved in genome organization and capsid stability. Taken together, these structures provide a new level of understanding for capsid stabilization and genome organization of segmented dsRNA viruses. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33403.map.gz emd_33403.map.gz | 706.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33403-v30.xml emd-33403-v30.xml emd-33403.xml emd-33403.xml | 23.2 KB 23.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_33403_fsc.xml emd_33403_fsc.xml | 28 KB | Display |  FSC data file FSC data file |
| Images |  emd_33403.png emd_33403.png | 296.2 KB | ||
| Filedesc metadata |  emd-33403.cif.gz emd-33403.cif.gz | 6.9 KB | ||
| Others |  emd_33403_half_map_1.map.gz emd_33403_half_map_1.map.gz emd_33403_half_map_2.map.gz emd_33403_half_map_2.map.gz | 706.8 MB 706.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33403 http://ftp.pdbj.org/pub/emdb/structures/EMD-33403 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33403 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33403 | HTTPS FTP |
-Related structure data
| Related structure data |  7xr2MC  7xr3C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_33403.map.gz / Format: CCP4 / Size: 1.9 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33403.map.gz / Format: CCP4 / Size: 1.9 GB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_33403_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
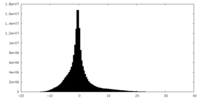
| Density Histograms |
-Half map: #1
| File | emd_33403_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
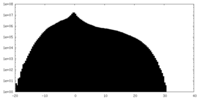
| Density Histograms |
- Sample components
Sample components
-Entire : Scylla serrata reovirus SZ-2007
| Entire | Name:  Scylla serrata reovirus SZ-2007 Scylla serrata reovirus SZ-2007 |
|---|---|
| Components |
|
-Supramolecule #1: Scylla serrata reovirus SZ-2007
| Supramolecule | Name: Scylla serrata reovirus SZ-2007 / type: virus / ID: 1 / Parent: 0 / Macromolecule list: all / NCBI-ID: 458682 / Sci species name: Scylla serrata reovirus SZ-2007 / Virus type: VIRION / Virus isolate: STRAIN / Virus enveloped: No / Virus empty: No |
|---|---|
| Host (natural) | Organism:  Scylla serrata (giant mud crab) Scylla serrata (giant mud crab) |
-Macromolecule #1: VP3
| Macromolecule | Name: VP3 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Scylla serrata reovirus SZ-2007 Scylla serrata reovirus SZ-2007 |
| Molecular weight | Theoretical: 97.019383 KDa |
| Sequence | String: MASTTRLVND RKQLEQQVKD DARILADARG LNITTVANDS ATGGQAIRNV GPNDEATIKA LDNVIKQIEA LSVIVNRSEK ADDAQILGP NTYKQLLEHL FSPEENVYIL LPIQAYTGGV IDRRDASFSN FAYSIASKLM MELSAATHNK IFTDYTRIAA S ALGPEIST ...String: MASTTRLVND RKQLEQQVKD DARILADARG LNITTVANDS ATGGQAIRNV GPNDEATIKA LDNVIKQIEA LSVIVNRSEK ADDAQILGP NTYKQLLEHL FSPEENVYIL LPIQAYTGGV IDRRDASFSN FAYSIASKLM MELSAATHNK IFTDYTRIAA S ALGPEIST EGMPLFSLIE SLELTEAETS RLPVIQDSMV IQKSTATVGN AQQGISTINI KRVPFVGSAF QQVIDQLLWE YS TTSLTTK EQRRQRITEM VNDRRIMIQK LTLAEKPQVM RHVTTEINND LFFKMSPVAQ LYIYHLDRAF LDGVGFTPLA EKQ QQLQLQ LKTNILTANL IRSAINGMNT ESNLEVAIKM MQAAQLHRAS IEIAFPMNVS LSPEIIVQCF IVWMSIPEQL LSDR SNFII AAVIWAGFSA DDSYADIMRR SARASDRQNY DIIKAALSSR KFKLPRASTT LFDENEPVVR RYQIGRVYAP FPVDR YGSP VYSNCTKVEL ASDYNAEGFT IRKDDFRALQ AVLRIDEDRA ADMFTTLRIM ISSIPAVWYD AEVVHYPHTA VELEQL AAY GLTGAYPRTN HSVDTIVKTV NNISATYSTI AQMLSTIDLD PTRYGTSESI DKFKIAWENV ESVLNMEGND FVKTIMY AY EDNFPKKDFY MMLKQIASDG QGAHPIAAAI DQLRTIVYRE PERFGYIDSV ILTHNPDVDT AYNRFFHLHP IVTNQPSN T IKNAQLWNEM RLEQQVEHIK AGPVRIIGPF HVTYNYLSEE EDMPATSHII MKDNMILNDH LTFNFVKRER RNNKKRVSS FRYKAVEMYV AVRISRFQLE VLRDLHDLVR SRTYLDVSKS PLATTPIRVV EYVR UniProtKB: VP3 |
-Macromolecule #2: VP11
| Macromolecule | Name: VP11 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Scylla serrata reovirus SZ-2007 Scylla serrata reovirus SZ-2007 |
| Molecular weight | Theoretical: 23.750701 KDa |
| Sequence | String: MNWSKAINFQ PFMLETRPPL TTIPIMDQLV EIGERSNQKW SMTDRLFFAI RKINPIFVTS SQIPSKFDYT ILQMPTQLIA SLKETLLFL AFSYYLREYQ DKVGQMKFYP VAMKNMIPIV NYLKDRVHNN FDTTLEQAYR QNVVHTLSAS DAFDLLSGMI A TTRLDLIQ ...String: MNWSKAINFQ PFMLETRPPL TTIPIMDQLV EIGERSNQKW SMTDRLFFAI RKINPIFVTS SQIPSKFDYT ILQMPTQLIA SLKETLLFL AFSYYLREYQ DKVGQMKFYP VAMKNMIPIV NYLKDRVHNN FDTTLEQAYR QNVVHTLSAS DAFDLLSGMI A TTRLDLIQ RTRICPELLN VLNKMSFILI YAPNRPSILS WKNQS UniProtKB: VP11 |
-Macromolecule #3: VP12
| Macromolecule | Name: VP12 / type: protein_or_peptide / ID: 3 / Number of copies: 13 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Scylla serrata reovirus SZ-2007 Scylla serrata reovirus SZ-2007 |
| Molecular weight | Theoretical: 29.730963 KDa |
| Sequence | String: MNLEINNFAP AISSIGSQLC SLSAQKLLTC RKQYGNGAKS FEEFYAEIGG IIGMMGINSQ TPSGIREAIY RLYQSAFLFG DIFPESFGI QNTQNIKPPP GFTAPAKKLE VVLPQGGAFD LIYNNGEIRV TTTRNVQAGD LVCTVTFPIQ GSVIATRNCH V NEIGGQLT ...String: MNLEINNFAP AISSIGSQLC SLSAQKLLTC RKQYGNGAKS FEEFYAEIGG IIGMMGINSQ TPSGIREAIY RLYQSAFLFG DIFPESFGI QNTQNIKPPP GFTAPAKKLE VVLPQGGAFD LIYNNGEIRV TTTRNVQAGD LVCTVTFPIQ GSVIATRNCH V NEIGGQLT TTRPEIIASV PMPARTVIVA SFDAIEIGYG EGDDLFAIGI AILSNRFNGQ ITPMSRHNYM TQMFANLPAN MS ERDSSAV LHFAQAAPVV LGMMERLTGA PKWVLDY UniProtKB: VP12 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 101.325 kPa |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Temperature | Min: 80.0 K / Max: 100.0 K |
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Detector mode: INTEGRATING / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Digitization - Frames/image: 1-16 / Number grids imaged: 1 / Number real images: 3595 / Average exposure time: 1.1 sec. / Average electron dose: 25.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Calibrated defocus max: 3.0 µm / Calibrated defocus min: 0.6 µm / Calibrated magnification: 128440 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.6 µm / Nominal magnification: 75000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)