+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-7133 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Human TRPM4 ion channel in lipid nanodiscs in a calcium-bound state | ||||||||||||||||||
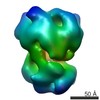
 マップデータ マップデータ | Sharpened map of human TRPM4 ion channel in lipid nanodisc with 5 mM CaCl2. This reconstruction is of the entire molecule with C4 symmetry. | ||||||||||||||||||
 試料 試料 |
| ||||||||||||||||||
 キーワード キーワード | TRPM4 / TRPM channel / TRP channel / MEMBRANE PROTEIN | ||||||||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報positive regulation of atrial cardiac muscle cell action potential / positive regulation of regulation of vascular associated smooth muscle cell membrane depolarization / sodium channel complex / regulation of T cell cytokine production / membrane depolarization during AV node cell action potential / membrane depolarization during bundle of His cell action potential / membrane depolarization during Purkinje myocyte cell action potential / negative regulation of bone mineralization / ligand-gated calcium channel activity / regulation of ventricular cardiac muscle cell action potential ...positive regulation of atrial cardiac muscle cell action potential / positive regulation of regulation of vascular associated smooth muscle cell membrane depolarization / sodium channel complex / regulation of T cell cytokine production / membrane depolarization during AV node cell action potential / membrane depolarization during bundle of His cell action potential / membrane depolarization during Purkinje myocyte cell action potential / negative regulation of bone mineralization / ligand-gated calcium channel activity / regulation of ventricular cardiac muscle cell action potential / sodium ion import across plasma membrane / calcium-activated cation channel activity / sodium channel activity / inorganic cation transmembrane transport / TRP channels / dendritic cell chemotaxis / cellular response to ATP / positive regulation of heart rate / regulation of heart rate by cardiac conduction / positive regulation of insulin secretion involved in cellular response to glucose stimulus / protein sumoylation / positive regulation of fat cell differentiation / negative regulation of osteoblast differentiation / positive regulation of vasoconstriction / positive regulation of adipose tissue development / calcium-mediated signaling / calcium ion transmembrane transport / Sensory perception of sweet, bitter, and umami (glutamate) taste / positive regulation of canonical Wnt signaling pathway / positive regulation of cytosolic calcium ion concentration / protein homotetramerization / adaptive immune response / calmodulin binding / neuronal cell body / calcium ion binding / positive regulation of cell population proliferation / Golgi apparatus / endoplasmic reticulum / nucleoplasm / ATP binding / identical protein binding / membrane / plasma membrane / cytosol 類似検索 - 分子機能 | ||||||||||||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||||||||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.1 Å | ||||||||||||||||||
 データ登録者 データ登録者 | Autzen HE / Myasnikov AG | ||||||||||||||||||
| 資金援助 |  米国, 米国,  デンマーク, 5件 デンマーク, 5件
| ||||||||||||||||||
 引用 引用 |  ジャーナル: Science / 年: 2018 ジャーナル: Science / 年: 2018タイトル: Structure of the human TRPM4 ion channel in a lipid nanodisc. 著者: Henriette E Autzen / Alexander G Myasnikov / Melody G Campbell / Daniel Asarnow / David Julius / Yifan Cheng /   要旨: Transient receptor potential (TRP) melastatin 4 (TRPM4) is a widely expressed cation channel associated with a variety of cardiovascular disorders. TRPM4 is activated by increased intracellular ...Transient receptor potential (TRP) melastatin 4 (TRPM4) is a widely expressed cation channel associated with a variety of cardiovascular disorders. TRPM4 is activated by increased intracellular calcium in a voltage-dependent manner but, unlike many other TRP channels, is permeable to monovalent cations only. Here we present two structures of full-length human TRPM4 embedded in lipid nanodiscs at ~3-angstrom resolution, as determined by single-particle cryo-electron microscopy. These structures, with and without calcium bound, reveal a general architecture for this major subfamily of TRP channels and a well-defined calcium-binding site within the intracellular side of the S1-S4 domain. The structures correspond to two distinct closed states. Calcium binding induces conformational changes that likely prime the channel for voltage-dependent opening. | ||||||||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_7133.map.gz emd_7133.map.gz | 4.2 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-7133-v30.xml emd-7133-v30.xml emd-7133.xml emd-7133.xml | 27 KB 27 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_7133.png emd_7133.png | 191.2 KB | ||
| Filedesc metadata |  emd-7133.cif.gz emd-7133.cif.gz | 6.9 KB | ||
| その他 |  emd_7133_additional_1.map.gz emd_7133_additional_1.map.gz emd_7133_additional_2.map.gz emd_7133_additional_2.map.gz emd_7133_additional_3.map.gz emd_7133_additional_3.map.gz emd_7133_half_map_1.map.gz emd_7133_half_map_1.map.gz emd_7133_half_map_2.map.gz emd_7133_half_map_2.map.gz | 94.6 MB 2.6 MB 2.5 MB 94.2 MB 94.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7133 http://ftp.pdbj.org/pub/emdb/structures/EMD-7133 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7133 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7133 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_7133_validation.pdf.gz emd_7133_validation.pdf.gz | 755.4 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_7133_full_validation.pdf.gz emd_7133_full_validation.pdf.gz | 755 KB | 表示 | |
| XML形式データ |  emd_7133_validation.xml.gz emd_7133_validation.xml.gz | 14.2 KB | 表示 | |
| CIF形式データ |  emd_7133_validation.cif.gz emd_7133_validation.cif.gz | 16.6 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7133 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7133 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7133 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7133 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  6bqvMC  7132C  6bqrC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | |
| 電子顕微鏡画像生データ |  EMPIAR-10127 (タイトル: Human TRPM4 ion channel in a lipid nanodisc in a calcium-bound state EMPIAR-10127 (タイトル: Human TRPM4 ion channel in a lipid nanodisc in a calcium-bound stateData size: 84.4 Data #1: particle stacks of TRPM4 particles post 2D clean-up [picked particles - multiframe - processed]) |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_7133.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_7133.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Sharpened map of human TRPM4 ion channel in lipid nanodisc with 5 mM CaCl2. This reconstruction is of the entire molecule with C4 symmetry. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.046 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-追加マップ: Unsharpened map of human TRPM4 ion channel in...
| ファイル | emd_7133_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Unsharpened map of human TRPM4 ion channel in lipid nanodisc with 5 mM CaCl2. This reconstruction is of the entire molecule with C4 symmetry. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
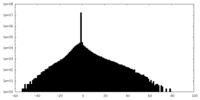
| 密度ヒストグラム |
-追加マップ: Sharpened map of human TRPM4 ion channel in...
| ファイル | emd_7133_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Sharpened map of human TRPM4 ion channel in lipid nanodisc with 5 mM CaCl2. This reconstruction is focused on transmembrane domain, central coiled-coil domain, and MHR4 domain. C4 symmetry is applied. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
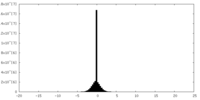
| 密度ヒストグラム |
-追加マップ: Sharpened map of human TRPM4 ion channel in...
| ファイル | emd_7133_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Sharpened map of human TRPM4 ion channel in lipid nanodisc with 5 mM CaCl2. This reconstruction is focused on monomeric soluble domain without symmetry. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
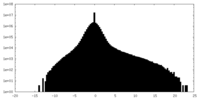
| 密度ヒストグラム |
-ハーフマップ: Human TRPM4 ion channel in lipid nanodiscs in...
| ファイル | emd_7133_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Human TRPM4 ion channel in lipid nanodiscs in a calcium-bound state, half map #1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
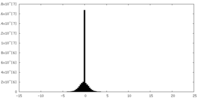
| 密度ヒストグラム |
-ハーフマップ: Half map #2 of human TRPM4 ion channel...
| ファイル | emd_7133_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map #2 of human TRPM4 ion channel in lipid nanodisc with 5 mM CaCl2. This reconstruction is of the entire molecule with C4 symmetry. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Human TRPM4 ion channel
| 全体 | 名称: Human TRPM4 ion channel |
|---|---|
| 要素 |
|
-超分子 #1: Human TRPM4 ion channel
| 超分子 | 名称: Human TRPM4 ion channel / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1 詳細: Human TRPM4 ion channel in lipid nanodiscs in a calcium-bound state |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 134.6 KDa |
-分子 #1: Transient receptor potential cation channel subfamily M member 4
| 分子 | 名称: Transient receptor potential cation channel subfamily M member 4 タイプ: protein_or_peptide / ID: 1 / コピー数: 4 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 134.770734 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: SGRSGVVPEK EQSWIPKIFK KKTCTTFIVD STDPGGTLCQ CGRPRTAHPA VAMEDAFGAA VVTVWDSDAH TTEKPTDAYG ELDFTGAGR KHSNFLRLSD RTDPAAVYSL VTRTWGFRAP NLVVSVLGGS GGPVLQTWLQ DLLRRGLVRA AQSTGAWIVT G GLHTGIGR ...文字列: SGRSGVVPEK EQSWIPKIFK KKTCTTFIVD STDPGGTLCQ CGRPRTAHPA VAMEDAFGAA VVTVWDSDAH TTEKPTDAYG ELDFTGAGR KHSNFLRLSD RTDPAAVYSL VTRTWGFRAP NLVVSVLGGS GGPVLQTWLQ DLLRRGLVRA AQSTGAWIVT G GLHTGIGR HVGVAVRDHQ MASTGGTKVV AMGVAPWGVV RNRDTLINPK GSFPARYRWR GDPEDGVQFP LDYNYSAFFL VD DGTHGCL GGENRFRLRL ESYISQQKTG VGGTGIDIPV LLLLIDGDEK MLTRIENATQ AQLPCLLVAG SGGAADCLAE TLE DTLAPG SGGARQGEAR DRIRRFFPKG DLEVLQAQVE RIMTRKELLT VYSSEDGSEE FETIVLKALV KACGSSEASA YLDE LRLAV AWNRVDIAQS ELFRGDIQWR SFHLEASLMD ALLNDRPEFV RLLISHGLSL GHFLTPMRLA QLYSAAPSNS LIRNL LDQA SHSAGTKAPA LKGGAAELRP PDVGHVLRML LGKMCAPRYP SGGAWDPHPG QGFGESMYLL SDKATSPLSL DAGLGQ APW SDLLLWALLL NRAQMAMYFW EMGSNAVSSA LGACLLLRVM ARLEPDAEEA ARRKDLAFKF EGMGVDLFGE CYRSSEV RA ARLLLRRCPL WGDATCLQLA MQADARAFFA QDGVQSLLTQ KWWGDMASTT PIWALVLAFF CPPLIYTRLI TFRKSEEE P TREELEFDMD SVINGEGPVG TADPAEKTPL GVPRQSGRPG CCGGRCGGRR CLRRWFHFWG APVTIFMGNV VSYLLFLLL FSRVLLVDFQ PAPPGSLELL LYFWAFTLLC EELRQGLSGG GGSLASGGPG PGHASLSQRL RLYLADSWNQ CDLVALTCFL LGVGCRLTP GLYHLGRTVL CIDFMVFTVR LLHIFTVNKQ LGPKIVIVSK MMKDVFFFLF FLGVWLVAYG VATEGLLRPR D SDFPSILR RVFYRPYLQI FGQIPQEDMD VALMEHSNCS SEPGFWAHPP GAQAGTCVSQ YANWLVVLLL VIFLLVANIL LV NLLIAMF SYTFGKVQGN SDLYWKAQRY RLIREFHSRP ALAPPFIVIS HLRLLLRQLC RRPRSPQPSS PALEHFRVYL SKE AERKLL TWESVHKENF LLARARDKRE SDSERLKRTS QKVDLALKQL GHIREYEQRL KVLEREVQQC SRVLGWVAEA LSRS ALLPP GGPPPPDLPG SKD UniProtKB: Transient receptor potential cation channel subfamily M member 4 |
-分子 #2: CALCIUM ION
| 分子 | 名称: CALCIUM ION / タイプ: ligand / ID: 2 / コピー数: 4 / 式: CA |
|---|---|
| 分子量 | 理論値: 40.078 Da |
-分子 #3: CHOLESTEROL HEMISUCCINATE
| 分子 | 名称: CHOLESTEROL HEMISUCCINATE / タイプ: ligand / ID: 3 / コピー数: 12 / 式: Y01 |
|---|---|
| 分子量 | 理論値: 486.726 Da |
| Chemical component information |  ChemComp-Y01: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 2.2 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.4 構成要素:
| ||||||||||||
| グリッド | モデル: Quantifoil R1.2/1.3 / 材質: COPPER / メッシュ: 200 / 支持フィルム - 材質: CARBON / 支持フィルム - トポロジー: HOLEY | ||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 283 K / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: SUPER-RESOLUTION / 撮影したグリッド数: 1 / 実像数: 2954 / 平均露光時間: 12.0 sec. / 平均電子線量: 62.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 1.5 µm / 最小 デフォーカス(公称値): 0.5 µm / 倍率(公称値): 22500 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 精密化 | プロトコル: AB INITIO MODEL |
|---|---|
| 得られたモデル |  PDB-6bqv: |
 ムービー
ムービー コントローラー
コントローラー








 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)