[English] 日本語
 Yorodumi
Yorodumi- PDB-6avo: Cryo-EM structure of human immunoproteasome with a novel noncompe... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6avo | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human immunoproteasome with a novel noncompetitive inhibitor that selectively inhibits activated lymphocytes | |||||||||||||||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||||||||||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / inhibitor / complex / immunoproteasome / HYDROLASE-HYDROLASE INHIBITOR complex | |||||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationspermatoproteasome complex / antigen processing and presentation of exogenous peptide antigen via MHC class I, TAP-dependent / purine ribonucleoside triphosphate binding / Antigen processing: Ub, ATP-independent proteasomal degradation / sperm glycocalyx / Regulation of ornithine decarboxylase (ODC) / Proteasome assembly / antigen processing and presentation / proteasome core complex / perinuclear theca ...spermatoproteasome complex / antigen processing and presentation of exogenous peptide antigen via MHC class I, TAP-dependent / purine ribonucleoside triphosphate binding / Antigen processing: Ub, ATP-independent proteasomal degradation / sperm glycocalyx / Regulation of ornithine decarboxylase (ODC) / Proteasome assembly / antigen processing and presentation / proteasome core complex / perinuclear theca / Cross-presentation of soluble exogenous antigens (endosomes) / Somitogenesis / humoral immune response / myofibril / proteasomal ubiquitin-independent protein catabolic process / sperm head-tail coupling apparatus / T cell proliferation / immune system process / proteasome endopeptidase complex / NF-kappaB binding / proteasome core complex, beta-subunit complex / fat cell differentiation / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / : / ciliary tip / proteasome complex / Regulation of activated PAK-2p34 by proteasome mediated degradation / sarcomere / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / Asymmetric localization of PCP proteins / Ubiquitin-dependent degradation of Cyclin D / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / AUF1 (hnRNP D0) binds and destabilizes mRNA / TNFR2 non-canonical NF-kB pathway / centriole / sperm end piece / P-body / Assembly of the pre-replicative complex / negative regulation of inflammatory response to antigenic stimulus / Vpu mediated degradation of CD4 / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / lipopolysaccharide binding / Dectin-1 mediated noncanonical NF-kB signaling / Degradation of DVL / Degradation of AXIN / Degradation of CRY and PER proteins / Hh mutants are degraded by ERAD / Activation of NF-kappaB in B cells / G2/M Checkpoints / Degradation of GLI1 by the proteasome / Hedgehog ligand biogenesis / Autodegradation of the E3 ubiquitin ligase COP1 / Regulation of RUNX3 expression and activity / Defective CFTR causes cystic fibrosis / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 / Negative regulation of NOTCH4 signaling / APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 / Hedgehog 'on' state / Vif-mediated degradation of APOBEC3G / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / Degradation of GLI2 by the proteasome / GLI3 is processed to GLI3R by the proteasome / MAPK6/MAPK4 signaling / Ubiquitin-Mediated Degradation of Phosphorylated Cdc25A / Degradation of CDH1 / Degradation of beta-catenin by the destruction complex / Oxygen-dependent proline hydroxylation of Hypoxia-inducible Factor Alpha / CDK-mediated phosphorylation and removal of Cdc6 / ABC-family protein mediated transport / CLEC7A (Dectin-1) signaling / SCF(Skp2)-mediated degradation of p27/p21 / FCERI mediated NF-kB activation / cell morphogenesis / response to virus / Regulation of expression of SLITs and ROBOs / Regulation of PTEN stability and activity / nuclear matrix / Interleukin-1 signaling / Orc1 removal from chromatin / Regulation of RAS by GAPs / Interferon alpha/beta signaling / Regulation of RUNX2 expression and activity / The role of GTSE1 in G2/M progression after G2 checkpoint / Separation of Sister Chromatids / UCH proteinases / KEAP1-NFE2L2 pathway / Downstream TCR signaling / Antigen processing: Ubiquitination & Proteasome degradation / sperm principal piece / RUNX1 regulates transcription of genes involved in differentiation of HSCs / ER-Phagosome pathway / Neddylation / regulation of inflammatory response / sperm midpiece / secretory granule lumen / endopeptidase activity / ficolin-1-rich granule lumen Similarity search - Function | |||||||||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||||||||||||||||||||||||||
 Authors Authors | Li, H. / Santos, R. / Bai, L. | |||||||||||||||||||||||||||||||||
| Funding support |  United States, 1items United States, 1items
| |||||||||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2017 Journal: Nat Commun / Year: 2017Title: Structure of human immunoproteasome with a reversible and noncompetitive inhibitor that selectively inhibits activated lymphocytes. Authors: Ruda de Luna Almeida Santos / Lin Bai / Pradeep K Singh / Naoka Murakami / Hao Fan / Wenhu Zhan / Yingrong Zhu / Xiuju Jiang / Kaiming Zhang / Jean Pierre Assker / Carl F Nathan / Huilin Li ...Authors: Ruda de Luna Almeida Santos / Lin Bai / Pradeep K Singh / Naoka Murakami / Hao Fan / Wenhu Zhan / Yingrong Zhu / Xiuju Jiang / Kaiming Zhang / Jean Pierre Assker / Carl F Nathan / Huilin Li / Jamil Azzi / Gang Lin /  Abstract: Proteasome inhibitors benefit patients with multiple myeloma and B cell-dependent autoimmune disorders but exert toxicity from inhibition of proteasomes in other cells. Toxicity should be minimized ...Proteasome inhibitors benefit patients with multiple myeloma and B cell-dependent autoimmune disorders but exert toxicity from inhibition of proteasomes in other cells. Toxicity should be minimized by reversible inhibition of the immunoproteasome β5i subunit while sparing the constitutive β5c subunit. Here we report β5i-selective inhibition by asparagine-ethylenediamine (AsnEDA)-based compounds and present the high-resolution cryo-EM structural analysis of the human immunoproteasome. Despite inhibiting noncompetitively, an AsnEDA inhibitor binds the active site. Hydrophobic interactions are accompanied by hydrogen bonding with β5i and β6 subunits. The inhibitors are far more cytotoxic for myeloma and lymphoma cell lines than for hepatocarcinoma or non-activated lymphocytes. They block human B-cell proliferation and promote apoptotic cell death selectively in antibody-secreting B cells, and to a lesser extent in activated human T cells. Reversible, β5i-selective inhibitors may be useful for treatment of diseases involving activated or neoplastic B cells or activated T cells. | |||||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6avo.cif.gz 6avo.cif.gz | 1 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6avo.ent.gz pdb6avo.ent.gz | 856.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6avo.json.gz 6avo.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/av/6avo https://data.pdbj.org/pub/pdb/validation_reports/av/6avo ftp://data.pdbj.org/pub/pdb/validation_reports/av/6avo ftp://data.pdbj.org/pub/pdb/validation_reports/av/6avo | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7010MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Proteasome subunit beta type- ... , 7 types, 14 molecules AFBECDSXTVUYWa
| #1: Protein | Mass: 21295.000 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P28065, proteasome endopeptidase complex #2: Protein | Mass: 24674.248 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P40306, proteasome endopeptidase complex #3: Protein | Mass: 22685.633 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P28062, proteasome endopeptidase complex #11: Protein | Mass: 23578.986 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P20618, proteasome endopeptidase complex #12: Protein | Mass: 22864.277 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P49721, proteasome endopeptidase complex #13: Protein | Mass: 22972.896 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P49720, proteasome endopeptidase complex #14: Protein | Mass: 24414.740 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P28070, proteasome endopeptidase complex |
|---|
-Proteasome subunit alpha type- ... , 7 types, 14 molecules GLHMINJQKROZPb
| #4: Protein | Mass: 29595.627 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P25786, proteasome endopeptidase complex #5: Protein | Mass: 26435.977 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P28066, proteasome endopeptidase complex #6: Protein | Mass: 27929.891 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: O14818, proteasome endopeptidase complex #7: Protein | Mass: 28469.252 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P25788, proteasome endopeptidase complex #8: Protein | Mass: 27432.459 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P60900, proteasome endopeptidase complex #9: Protein | Mass: 29525.842 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P25789, proteasome endopeptidase complex #10: Protein | Mass: 25927.535 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P25787, proteasome endopeptidase complex |
|---|
-Non-polymers , 1 types, 2 molecules 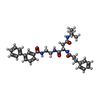
| #15: Chemical |
|---|
-Details
| Has protein modification | N |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: human immunoproteasome with a novel noncompetitive inhibitor that selectively inhibits activated lymphocytes Type: COMPLEX / Entity ID: #1-#14 / Source: NATURAL |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 8 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Microscopy | Model: JEOL 3200FS |
|---|---|
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 60 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.11.1_2575: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 75017 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj






