+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-6913 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | 4.9 Angstrom Cryo-EM structure of human mTOR complex 2 | |||||||||||||||
 マップデータ マップデータ | ||||||||||||||||
 試料 試料 |
| |||||||||||||||
 キーワード キーワード | Cryo-EM structure human mTORC2 / GENE REGULATION | |||||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報TORC2 signaling / regulation of peptidyl-serine phosphorylation / RNA polymerase III type 2 promoter sequence-specific DNA binding / RNA polymerase III type 1 promoter sequence-specific DNA binding / positive regulation of cytoplasmic translational initiation / T-helper 1 cell lineage commitment / positive regulation of pentose-phosphate shunt / regulation of locomotor rhythm / positive regulation of wound healing, spreading of epidermal cells / TORC2 complex ...TORC2 signaling / regulation of peptidyl-serine phosphorylation / RNA polymerase III type 2 promoter sequence-specific DNA binding / RNA polymerase III type 1 promoter sequence-specific DNA binding / positive regulation of cytoplasmic translational initiation / T-helper 1 cell lineage commitment / positive regulation of pentose-phosphate shunt / regulation of locomotor rhythm / positive regulation of wound healing, spreading of epidermal cells / TORC2 complex / cellular response to leucine starvation / TFIIIC-class transcription factor complex binding / regulation of membrane permeability / heart valve morphogenesis / negative regulation of lysosome organization / nucleus localization / RNA polymerase III type 3 promoter sequence-specific DNA binding / TORC1 complex / positive regulation of transcription of nucleolar large rRNA by RNA polymerase I / regulation of cellular response to oxidative stress / calcineurin-NFAT signaling cascade / regulation of osteoclast differentiation / voluntary musculoskeletal movement / TORC1 signaling / positive regulation of keratinocyte migration / phosphatidic acid binding / cellular response to L-leucine / Amino acids regulate mTORC1 / MTOR signalling / cellular response to nutrient / cellular response to methionine / Energy dependent regulation of mTOR by LKB1-AMPK / regulation of autophagosome assembly / energy reserve metabolic process / negative regulation of cell size / ruffle organization / negative regulation of Ras protein signal transduction / phosphatidylinositol-3,4-bisphosphate binding / cellular response to osmotic stress / phosphatidylinositol-3,5-bisphosphate binding / anoikis / cardiac muscle cell development / negative regulation of protein localization to nucleus / embryo development ending in birth or egg hatching / regulation of establishment of cell polarity / positive regulation of transcription by RNA polymerase III / negative regulation of calcineurin-NFAT signaling cascade / regulation of myelination / positive regulation of actin filament polymerization / regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / regulation of cell size / negative regulation of macroautophagy / positive regulation of oligodendrocyte differentiation / lysosome organization / Macroautophagy / positive regulation of myotube differentiation / behavioral response to pain / oligodendrocyte differentiation / Constitutive Signaling by AKT1 E17K in Cancer / mTORC1-mediated signalling / germ cell development / phosphatidylinositol-3,4,5-trisphosphate binding / CD28 dependent PI3K/Akt signaling / : / HSF1-dependent transactivation / positive regulation of TOR signaling / neuronal action potential / response to amino acid / TOR signaling / 'de novo' pyrimidine nucleobase biosynthetic process / endomembrane system / regulation of macroautophagy / positive regulation of translational initiation / cellular response to nutrient levels / positive regulation of lamellipodium assembly / phosphorylation / phagocytic vesicle / positive regulation of lipid biosynthetic process / heart morphogenesis / positive regulation of epithelial to mesenchymal transition / cardiac muscle contraction / regulation of cellular response to heat / phosphatidylinositol-4,5-bisphosphate binding / positive regulation of stress fiber assembly / cytoskeleton organization / positive regulation of endothelial cell proliferation / T cell costimulation / substantia nigra development / cellular response to starvation / cellular response to amino acid starvation / post-embryonic development / positive regulation of glycolytic process / protein serine/threonine kinase activator activity / negative regulation of autophagy / response to nutrient / response to nutrient levels / VEGFR2 mediated vascular permeability / regulation of signal transduction by p53 class mediator / Regulation of PTEN gene transcription / positive regulation of translation 類似検索 - 分子機能 | |||||||||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 4.9 Å | |||||||||||||||
 データ登録者 データ登録者 | Chen X / Liu M / Tian Y / Wang H / Wang J / Xu Y | |||||||||||||||
| 資金援助 |  中国, 4件 中国, 4件
| |||||||||||||||
 引用 引用 |  ジャーナル: Cell Res / 年: 2018 ジャーナル: Cell Res / 年: 2018タイトル: Cryo-EM structure of human mTOR complex 2. 著者: Xizi Chen / Mengjie Liu / Yuan Tian / Jiabei Li / Yilun Qi / Dan Zhao / Zihan Wu / Min Huang / Catherine C L Wong / Hong-Wei Wang / Jiawei Wang / Huirong Yang / Yanhui Xu /  要旨: Mechanistic target of rapamycin (mTOR) complex 2 (mTORC2) plays an essential role in regulating cell proliferation through phosphorylating AGC protein kinase family members, including AKT, PKC and ...Mechanistic target of rapamycin (mTOR) complex 2 (mTORC2) plays an essential role in regulating cell proliferation through phosphorylating AGC protein kinase family members, including AKT, PKC and SGK1. The functional core complex consists of mTOR, mLST8, and two mTORC2-specific components, Rictor and mSin1. Here we investigated the intermolecular interactions within mTORC2 complex and determined its cryo-electron microscopy structure at 4.9 Å resolution. The structure reveals a hollow rhombohedral fold with a 2-fold symmetry. The dimerized mTOR serves as a scaffold for the complex assembly. The N-terminal half of Rictor is composed of helical repeat clusters and binds to mTOR through multiple contacts. mSin1 is located close to the FRB domain and catalytic cavity of mTOR. Rictor and mSin1 together generate steric hindrance to inhibit binding of FKBP12-rapamycin to mTOR, revealing the mechanism for rapamycin insensitivity of mTORC2. The mTOR dimer in mTORC2 shows more compact conformation than that of mTORC1 (rapamycin sensitive), which might result from the interaction between mTOR and Rictor-mSin1. Structural comparison shows that binding of Rictor and Raptor (mTORC1-specific component) to mTOR is mutually exclusive. Our study provides a basis for understanding the assembly of mTORC2 and a framework to further characterize the regulatory mechanism of mTORC2 pathway. | |||||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_6913.map.gz emd_6913.map.gz | 18.8 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-6913-v30.xml emd-6913-v30.xml emd-6913.xml emd-6913.xml | 23.4 KB 23.4 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_6913.png emd_6913.png | 209.1 KB | ||
| Filedesc metadata |  emd-6913.cif.gz emd-6913.cif.gz | 8.6 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6913 http://ftp.pdbj.org/pub/emdb/structures/EMD-6913 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6913 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6913 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_6913_validation.pdf.gz emd_6913_validation.pdf.gz | 375.3 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_6913_full_validation.pdf.gz emd_6913_full_validation.pdf.gz | 374.9 KB | 表示 | |
| XML形式データ |  emd_6913_validation.xml.gz emd_6913_validation.xml.gz | 7.1 KB | 表示 | |
| CIF形式データ |  emd_6913_validation.cif.gz emd_6913_validation.cif.gz | 8 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6913 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6913 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6913 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-6913 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_6913.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_6913.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.01 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
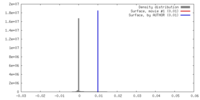
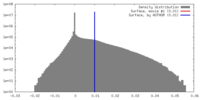
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : human mTOR Complex 2
| 全体 | 名称: human mTOR Complex 2 |
|---|---|
| 要素 |
|
-超分子 #1: human mTOR Complex 2
| 超分子 | 名称: human mTOR Complex 2 / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 1400 kDa/nm |
-分子 #1: Serine/threonine-protein kinase mTOR
| 分子 | 名称: Serine/threonine-protein kinase mTOR / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO / EC番号: non-specific serine/threonine protein kinase |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 289.257969 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MLGTGPAAAT TAATTSSNVS VLQQFASGLK SRNEETRAKA AKELQHYVTM ELREMSQEES TRFYDQLNHH IFELVSSSDA NERKGGILA IASLIGVEGG NATRIGRFAN YLRNLLPSND PVVMEMASKA IGRLAMAGDT FTAEYVEFEV KRALEWLGAD R NEGRRHAA ...文字列: MLGTGPAAAT TAATTSSNVS VLQQFASGLK SRNEETRAKA AKELQHYVTM ELREMSQEES TRFYDQLNHH IFELVSSSDA NERKGGILA IASLIGVEGG NATRIGRFAN YLRNLLPSND PVVMEMASKA IGRLAMAGDT FTAEYVEFEV KRALEWLGAD R NEGRRHAA VLVLRELAIS VPTFFFQQVQ PFFDNIFVAV WDPKQAIREG AVAALRACLI LTTQREPKEM QKPQWYRHTF EE AEKGFDE TLAKEKGMNR DDRIHGALLI LNELVRISSM EGERLREEME EITQQQLVHD KYCKDLMGFG TKPRHITPFT SFQ AVQPQQ SNALVGLLGY SSHQGLMGFG TSPSPAKSTL VESRCCRDLM EEKFDQVCQW VLKCRNSKNS LIQMTILNLL PRLA AFRPS AFTDTQYLQD TMNHVLSCVK KEKERTAAFQ ALGLLSVAVR SEFKVYLPRV LDIIRAALPP KDFAHKRQKA MQVDA TVFT CISMLARAMG PGIQQDIKEL LEPMLAVGLS PALTAVLYDL SRQIPQLKKD IQDGLLKMLS LVLMHKPLRH PGMPKG LAH QLASPGLTTL PEASDVGSIT LALRTLGSFE FEGHSLTQFV RHCADHFLNS EHKEIRMEAA RTCSRLLTPS IHLISGH AH VVSQTAVQVV ADVLSKLLVV GITDPDPDIR YCVLASLDER FDAHLAQAEN LQALFVALND QVFEIRELAI CTVGRLSS M NPAFVMPFLR KMLIQILTEL EHSGIGRIKE QSARMLGHLV SNAPRLIRPY MEPILKALIL KLKDPDPDPN PGVINNVLA TIGELAQVSG LEMRKWVDEL FIIIMDMLQD SSLLAKRQVA LWTLGQLVAS TGYVVEPYRK YPTLLEVLLN FLKTEQNQGT RREAIRVLG LLGALDPYKH KVNIGMIDQS RDASAVSLSE SKSSQDSSDY STSEMLVNMG NLPLDEFYPA VSMVALMRIF R DQSLSHHH TMVVQAITFI FKSLGLKCVQ FLPQVMPTFL NVIRVCDGAI REFLFQQLGM LVSFVKSHIR PYMDEIVTLM RE FWVMNTS IQSTIILLIE QIVVALGGEF KLYLPQLIPH MLRVFMHDNS PGRIVSIKLL AAIQLFGANL DDYLHLLLPP IVK LFDAPE APLPSRKAAL ETVDRLTESL DFTDYASRII HPIVRTLDQS PELRSTAMDT LSSLVFQLGK KYQIFIPMVN KVLV RHRIN HQRYDVLICR IVKGYTLADE EEDPLIYQHR MLRSGQGDAL ASGPVETGPM KKLHVSTINL QKAWGAARRV SKDDW LEWL RRLSLELLKD SSSPSLRSCW ALAQAYNPMA RDLFNAAFVS CWSELNEDQQ DELIRSIELA LTSQDIAEVT QTLLNL AEF MEHSDKGPLP LRDDNGIVLL GERAAKCRAY AKALHYKELE FQKGPTPAIL ESLISINNKL QQPEAAAGVL EYAMKHF GE LEIQATWYEK LHEWEDALVA YDKKMDTNKD DPELMLGRMR CLEALGEWGQ LHQQCCEKWT LVNDETQAKM ARMAAAAA W GLGQWDSMEE YTCMIPRDTH DGAFYRAVLA LHQDLFSLAQ QCIDKARDLL DAELTAMAGE SYSRAYGAMV SCHMLSELE EVIQYKLVPE RREIIRQIWW ERLQGCQRIV EDWQKILMVR SLVVSPHEDM RTWLKYASLC GKSGRLALAH KTLVLLLGVD PSRQLDHPL PTVHPQVTYA YMKNMWKSAR KIDAFQHMQH FVQTMQQQAQ HAIATEDQQH KQELHKLMAR CFLKLGEWQL N LQGINEST IPKVLQYYSA ATEHDRSWYK AWHAWAVMNF EAVLHYKHQN QARDEKKKLR HASGANITNA TTAATTAATA TT TASTEGS NSESEAESTE NSPTPSPLQK KVTEDLSKTL LMYTVPAVQG FFRSISLSRG NNLQDTLRVL TLWFDYGHWP DVN EALVEG VKAIQIDTWL QVIPQLIARI DTPRPLVGRL IHQLLTDIGR YHPQALIYPL TVASKSTTTA RHNAANKILK NMCE HSNTL VQQAMMVSEE LIRVAILWHE MWHEGLEEAS RLYFGERNVK GMFEVLEPLH AMMERGPQTL KETSFNQAYG RDLME AQEW CRKYMKSGNV KDLTQAWDLY YHVFRRISKQ LPQLTSLELQ YVSPKLLMCR DLELAVPGTY DPNQPIIRIQ SIAPSL QVI TSKQRPRKLT LMGSNGHEFV FLLKGHEDLR QDERVMQLFG LVNTLLANDP TSLRKNLSIQ RYAVIPLSTN SGLIGWV PH CDTLHALIRD YREKKKILLN IEHRIMLRMA PDYDHLTLMQ KVEVFEHAVN NTAGDDLAKL LWLKSPSSEV WFDRRTNY T RSLAVMSMVG YILGLGDRHP SNLMLDRLSG KILHIDFGDC FEVAMTREKF PEKIPFRLTR MLTNAMEVTG LDGNYRITC HTVMEVLREH KDSVMAVLEA FVYDPLLNWR LMDTNTKGNK RSRTRTDSYS AGQSVEILDG VELGEPAHKK TGTTVPESIH SFIGDGLVK PEALNKKAIQ IINRVRDKLT GRDFSHDDTL DVPTQVELLI KQATSHENLC QCYIGWCPFW UniProtKB: Serine/threonine-protein kinase mTOR |
-分子 #2: Target of rapamycin complex subunit LST8
| 分子 | 名称: Target of rapamycin complex subunit LST8 / タイプ: protein_or_peptide / ID: 2 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 35.91009 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MNTSPGTVGS DPVILATAGY DHTVRFWQAH SGICTRTVQH QDSQVNALEV TPDRSMIAAA GYQHIRMYDL NSNNPNPIIS YDGVNKNIA SVGFHEDGRW MYTGGEDCTA RIWDLRSRNL QCQRIFQVNA PINCVCLHPN QAELIVGDQS GAIHIWDLKT D HNEQLIPE ...文字列: MNTSPGTVGS DPVILATAGY DHTVRFWQAH SGICTRTVQH QDSQVNALEV TPDRSMIAAA GYQHIRMYDL NSNNPNPIIS YDGVNKNIA SVGFHEDGRW MYTGGEDCTA RIWDLRSRNL QCQRIFQVNA PINCVCLHPN QAELIVGDQS GAIHIWDLKT D HNEQLIPE PEVSITSAHI DPDASYMAAV NSTGNCYVWN LTGGIGDEVT QLIPKTKIPA HTRYALQCRF SPDSTLLATC SA DQTCKIW RTSNFSLMTE LSIKSGNPGE SSRGWMWGCA FSGDSQYIVT ASSDNLARLW CVETGEIKRE YGGHQKAVVC LAF NDSVLG UniProtKB: Target of rapamycin complex subunit LST8 |
-分子 #3: Rapamycin-insensitive companion of mTOR
| 分子 | 名称: Rapamycin-insensitive companion of mTOR / タイプ: protein_or_peptide / ID: 3 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 175.1425 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MAAIGRGRSL KNLRVRGRND SGEENVPLDL TREPSDNLRE ILQNVARLQG VSNMRKLGHL NNFTKLLCDI GHSEEKLGFH YEDIIICLR LALLNEAKEV RAAGLRALRY LIQDSSILQK VLKLKVDYLI ARCIDIQQSN EVERTQALRL VRKMITVNAS L FPSSVTNS ...文字列: MAAIGRGRSL KNLRVRGRND SGEENVPLDL TREPSDNLRE ILQNVARLQG VSNMRKLGHL NNFTKLLCDI GHSEEKLGFH YEDIIICLR LALLNEAKEV RAAGLRALRY LIQDSSILQK VLKLKVDYLI ARCIDIQQSN EVERTQALRL VRKMITVNAS L FPSSVTNS LIAVGNDGLQ ERDRMVRACI AIICELALQN PEVVALRGGL NTILKNVIDC QLSRINEALI TTILHLLNHP KT RQYVRAD VELERILAPY TDFHYRHSPD TAEGQLKEDR EARFLASKMG IIATFRSWAG IINLCKPGNS GIQSLIGVLC IPN MEIRRG LLEVLYDIFR LPLPVVTEEF IEALLSVDPG RFQDSWRLSD GFVAAEAKTI LPHRARSRPD LMDNYLALIL SAFI RNGLL EGLVEVITNS DDHISVRATI LLGELLHMAN TILPHSHSHH LHCLPTLMNM AASFDIPKEK RLRASAALNC LKRFH EMKK RGPKPYSLHL DHIIQKAIAT HQKRDQYLRV QKDIFILKDT EEALLINLRD SQVLQHKENL EWNWNLIGTI LKWPNV NLR NYKDEQLHRF VRRLLYFYKP SSKLYANLDL DFAKAKQLTV VGCQFTEFLL ESEEDGQGYL EDLVKDIVQW LNASSGM KP ERSLQNNGLL TTLSQHYFLF IGTLSCHPHG VKMLEKCSVF QCLLNLCSLK NQDHLLKLTV SSLDYSRDGL ARVILSKI L TAATDACRLY ATKHLRVLLR ANVEFFNNWG IELLVTQLHD KNKTISSEAL DILDEACEDK ANLHALIQMK PALSHLGDK GLLLLLRFLS IPKGFSYLNE RGYVAKQLEK WHREYNSKYV DLIEEQLNEA LTTYRKPVDG DNYVRRSNQR LQRPHVYLPI HLYGQLVHH KTGCHLLEVQ NIITELCRNV RTPDLDKWEE IKKLKASLWA LGNIGSSNWG LNLLQEENVI PDILKLAKQC E VLSIRGTC VYVLGLIAKT KQGCDILKCH NWDAVRHSRK HLWPVVPDDV EQLCNELSSI (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) UniProtKB: Rapamycin-insensitive companion of mTOR |
-分子 #4: Target of rapamycin complex 2 subunit MAPKAP1
| 分子 | 名称: Target of rapamycin complex 2 subunit MAPKAP1 / タイプ: protein_or_peptide / ID: 4 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 55.158477 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) ...文字列: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)IL SVRLEQCPLQ LNNPFNEY S KFDGKGHVGT TATKKIDVYL PLHSSQDRLL PMTVVTMASA RVQDLIGLIC WQYTSEGREP KLNDNVSAYC LHIAEDDGE VDTDFPPLDS NEPIHKFGFS TLALVEKYSS PGLTSKESLF VRINAAHGFS LIQVDNTKVT MKEILLKAVK RRKGSQKVSG PQYRLEKQS EPNVAVDLDS TLESQSAWEF CLVRENSSRA DGVFEEDSQI DIATVQDMLS SHHYKSFKVS MIHRLRFTTD V QLGISGDK VEIDPVTNQK ASTKFWIKQK PISIDSDLLC ACDLAEEKSP SHAIFKLTYL SNHDYKHLYF ESDAATVNEI VL KVNYILE SRASTARADY FAQKQRKLNR RTSFSFQKEK KSGQQ UniProtKB: Target of rapamycin complex 2 subunit MAPKAP1 |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 1.5 mg/mL |
|---|---|
| 緩衝液 | pH: 7.4 |
| グリッド | モデル: Quantifoil R1.2/1.3 / 材質: GOLD / メッシュ: 300 / 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 雰囲気: AIR |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 282 K / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: COUNTING / デジタル化 - 画像ごとのフレーム数: 1-32 / 平均露光時間: 8.0 sec. / 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 100.0 µm / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 初期モデル | モデルのタイプ: EMDB MAP |
|---|---|
| 最終 再構成 | 想定した対称性 - 点群: C2 (2回回転対称) / 解像度のタイプ: BY AUTHOR / 解像度: 4.9 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: RELION (ver. 2.0) / 使用した粒子像数: 195353 |
| 初期 角度割当 | タイプ: OTHER |
| 最終 角度割当 | タイプ: ANGULAR RECONSTITUTION / ソフトウェア - 名称: RELION (ver. 2.0) |
-原子モデル構築 1
| 精密化 | 空間: REAL / プロトコル: OTHER |
|---|---|
| 得られたモデル |  PDB-5zcs: |
 ムービー
ムービー コントローラー
コントローラー






















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)