[English] 日本語
 Yorodumi
Yorodumi- PDB-5dlt: Crystal structure of Autotaxin (ENPP2) with 7-alpha-hydroxycholesterol -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5dlt | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Autotaxin (ENPP2) with 7-alpha-hydroxycholesterol | |||||||||
 Components Components | Ectonucleotide pyrophosphatase/phosphodiesterase family member 2 | |||||||||
 Keywords Keywords | HYDROLASE / Autotaxin / ENPP2 / LPA / steroids / bile salts | |||||||||
| Function / homology |  Function and homology information Function and homology informationresponse to polycyclic arene / alkylglycerophosphoethanolamine phosphodiesterase / sphingolipid catabolic process / phospholipase D / phosphatidylcholine catabolic process / phospholipid catabolic process / D-type glycerophospholipase activity / positive regulation of lamellipodium morphogenesis / phosphodiesterase I activity / phosphatidylcholine lysophospholipase A1 activity ...response to polycyclic arene / alkylglycerophosphoethanolamine phosphodiesterase / sphingolipid catabolic process / phospholipase D / phosphatidylcholine catabolic process / phospholipid catabolic process / D-type glycerophospholipase activity / positive regulation of lamellipodium morphogenesis / phosphodiesterase I activity / phosphatidylcholine lysophospholipase A1 activity / scavenger receptor activity / alkylglycerophosphoethanolamine phosphodiesterase activity / cellular response to cadmium ion / polysaccharide binding / positive regulation of oligodendrocyte differentiation / positive regulation of epithelial cell migration / negative regulation of cell-matrix adhesion / positive regulation of focal adhesion assembly / estrous cycle / phospholipid metabolic process / positive regulation of substrate adhesion-dependent cell spreading / regulation of cell migration / cell chemotaxis / cellular response to estradiol stimulus / nucleic acid binding / immune response / calcium ion binding / positive regulation of cell population proliferation / : / zinc ion binding / membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.6 Å MOLECULAR REPLACEMENT / Resolution: 1.6 Å | |||||||||
 Authors Authors | Hausmann, J. / Joosten, R.P. / Perrakis, A. | |||||||||
| Funding support |  Netherlands, 2items Netherlands, 2items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2016 Journal: Nat Commun / Year: 2016Title: Steroid binding to Autotaxin links bile salts and lysophosphatidic acid signalling. Authors: Keune, W.J. / Hausmann, J. / Bolier, R. / Tolenaars, D. / Kremer, A. / Heidebrecht, T. / Joosten, R.P. / Sunkara, M. / Morris, A.J. / Matas-Rico, E. / Moolenaar, W.H. / Oude Elferink, R.P. / Perrakis, A. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5dlt.cif.gz 5dlt.cif.gz | 359.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5dlt.ent.gz pdb5dlt.ent.gz | 284.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5dlt.json.gz 5dlt.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dl/5dlt https://data.pdbj.org/pub/pdb/validation_reports/dl/5dlt ftp://data.pdbj.org/pub/pdb/validation_reports/dl/5dlt ftp://data.pdbj.org/pub/pdb/validation_reports/dl/5dlt | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5dlvC  5dlwC  2xr9S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Sugars , 2 types, 2 molecules A
| #1: Protein | Mass: 95117.641 Da / Num. of mol.: 1 / Mutation: N410A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) Homo sapiens (human)References: UniProt: Q64610, alkylglycerophosphoethanolamine phosphodiesterase |
|---|---|
| #2: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source |
-Non-polymers , 8 types, 560 molecules 
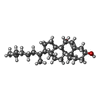













| #3: Chemical | | #4: Chemical | ChemComp-5JK / | #5: Chemical | ChemComp-CA / | #6: Chemical | ChemComp-IOD / #7: Chemical | ChemComp-SCN / #8: Chemical | #9: Chemical | ChemComp-GOL / #10: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.35 Å3/Da / Density % sol: 47.6 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6 Details: PEG 3350, 0.1M ammonium iodide 0.3M sodium thiocyanate, 23.5mg/ml heparin sulfate |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 0.97935 Å / Beamline: ID29 / Wavelength: 0.97935 Å | |||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 6M-F / Detector: PIXEL / Date: Aug 30, 2011 | |||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.97935 Å / Relative weight: 1 | |||||||||||||||||||||||||||
| Reflection | Redundancy: 2 % / Number: 205646 / Rmerge(I) obs: 0.065 / D res high: 1.6 Å / D res low: 44.02 Å / Num. obs: 105202 / % possible obs: 92.1 | |||||||||||||||||||||||||||
| Diffraction reflection shell |
| |||||||||||||||||||||||||||
| Reflection | Resolution: 1.6→44.02 Å / Num. obs: 105202 / % possible obs: 92.1 % / Redundancy: 2 % / Biso Wilson estimate: 29.61 Å2 / CC1/2: 0.997 / Rmerge(I) obs: 0.065 / Rpim(I) all: 0.061 / Net I/σ(I): 6.7 / Num. measured all: 205646 | |||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1 / Rejects: _
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2xr9 Resolution: 1.6→44.02 Å / Cor.coef. Fo:Fc: 0.97 / Cor.coef. Fo:Fc free: 0.962 / Matrix type: sparse / WRfactor Rfree: 0.192 / WRfactor Rwork: 0.166 / SU B: 4.722 / SU ML: 0.076 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.091 / ESU R Free: 0.088 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS U VALUES : WITH TLS ADDED
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.7 Å / Shrinkage radii: 0.7 Å / VDW probe radii: 1 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 103.32 Å2 / Biso mean: 28.789 Å2 / Biso min: 11.55 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 1.6→44.02 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj








