+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Synaptic Vesicle V-ATPase with synaptophysin and SidK, State 2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Membrane / Native / Complex / Synaptic / PROTON TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of opioid receptor signaling pathway / Metabolism of Angiotensinogen to Angiotensins / Transferrin endocytosis and recycling / Ion channel transport / Amino acids regulate mTORC1 / RHOA GTPase cycle / Insulin receptor recycling / eye pigmentation / central nervous system maturation / transporter activator activity ...regulation of opioid receptor signaling pathway / Metabolism of Angiotensinogen to Angiotensins / Transferrin endocytosis and recycling / Ion channel transport / Amino acids regulate mTORC1 / RHOA GTPase cycle / Insulin receptor recycling / eye pigmentation / central nervous system maturation / transporter activator activity / negative regulation of autophagic cell death / rostrocaudal neural tube patterning / cellular response to increased oxygen levels / positive regulation of transforming growth factor beta1 production / proton-transporting V-type ATPase, V1 domain / synaptic vesicle lumen acidification / endosome to plasma membrane protein transport / intracellular organelle / proton-transporting V-type ATPase, V0 domain / extrinsic component of synaptic vesicle membrane / P-type proton-exporting transporter activity / plasma membrane proton-transporting V-type ATPase complex / lysosomal lumen acidification / clathrin-coated vesicle membrane / endosomal lumen acidification / vacuolar proton-transporting V-type ATPase, V0 domain / vacuolar proton-transporting V-type ATPase, V1 domain / vacuolar transport / proton-transporting V-type ATPase complex / neuron spine / head morphogenesis / protein localization to cilium / regulation of short-term neuronal synaptic plasticity / neuron projection terminus / vacuolar proton-transporting V-type ATPase complex / dendritic spine membrane / regulation of cellular pH / syntaxin-1 binding / vacuolar acidification / osteoclast development / ROS and RNS production in phagocytes / Neutrophil degranulation / regulation of synaptic vesicle exocytosis / : / ATPase complex / cholesterol binding / presynaptic active zone / regulation of neuronal synaptic plasticity / autophagosome membrane / response to amyloid-beta / microvillus / regulation of MAPK cascade / ATPase activator activity / excitatory synapse / synaptic vesicle endocytosis / positive regulation of Wnt signaling pathway / cilium assembly / transmembrane transporter complex / regulation of macroautophagy / angiotensin maturation / axon terminus / ATP metabolic process / H+-transporting two-sector ATPase / proton transmembrane transport / ruffle / RNA endonuclease activity / phagocytic vesicle / proton-transporting ATPase activity, rotational mechanism / endoplasmic reticulum-Golgi intermediate compartment membrane / SH2 domain binding / proton-transporting ATP synthase activity, rotational mechanism / SNARE binding / receptor-mediated endocytosis / secretory granule / regulation of long-term neuronal synaptic plasticity / Schaffer collateral - CA1 synapse / neuromuscular junction / terminal bouton / transmembrane transport / synaptic vesicle membrane / small GTPase binding / cilium / endocytosis / melanosome / positive regulation of canonical Wnt signaling pathway / synaptic vesicle / presynapse / apical part of cell / presynaptic membrane / signaling receptor activity / cell body / ATPase binding / postsynaptic membrane / intracellular iron ion homeostasis / receptor-mediated endocytosis of virus by host cell / postsynaptic density / positive regulation of ERK1 and ERK2 cascade / lysosome / early endosome / endosome membrane Similarity search - Function | |||||||||
| Biological species |  | |||||||||
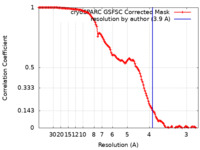
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Coupland CE / Rubinstein JL | |||||||||
| Funding support |  Canada, 1 items Canada, 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: High-resolution electron cryomicroscopy of V-ATPase in native synaptic vesicles. Authors: Claire E Coupland / Ryan Karimi / Stephanie A Bueler / Yingke Liang / Gautier M Courbon / Justin M Di Trani / Cassandra J Wong / Rayan Saghian / Ji-Young Youn / Lu-Yang Wang / John L Rubinstein /  Abstract: Intercellular communication in the nervous system occurs through the release of neurotransmitters into the synaptic cleft between neurons. In the presynaptic neuron, the proton pumping vesicular- or ...Intercellular communication in the nervous system occurs through the release of neurotransmitters into the synaptic cleft between neurons. In the presynaptic neuron, the proton pumping vesicular- or vacuolar-type ATPase (V-ATPase) powers neurotransmitter loading into synaptic vesicles (SVs), with the V complex dissociating from the membrane region of the enzyme before exocytosis. We isolated SVs from rat brain using SidK, a V-ATPase-binding bacterial effector protein. Single-particle electron cryomicroscopy allowed high-resolution structure determination of V-ATPase within the native SV membrane. In the structure, regularly spaced cholesterol molecules decorate the enzyme's rotor and the abundant SV protein synaptophysin binds the complex stoichiometrically. ATP hydrolysis during vesicle loading results in a loss of the V region of V-ATPase from the SV membrane, suggesting that loading is sufficient to induce dissociation of the enzyme. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_44354.map.gz emd_44354.map.gz | 204 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-44354-v30.xml emd-44354-v30.xml emd-44354.xml emd-44354.xml | 13.1 KB 13.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_44354_fsc.xml emd_44354_fsc.xml | 12.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_44354.png emd_44354.png | 118.7 KB | ||
| Filedesc metadata |  emd-44354.cif.gz emd-44354.cif.gz | 3.8 KB | ||
| Others |  emd_44354_half_map_1.map.gz emd_44354_half_map_1.map.gz emd_44354_half_map_2.map.gz emd_44354_half_map_2.map.gz | 200.6 MB 200.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-44354 http://ftp.pdbj.org/pub/emdb/structures/EMD-44354 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-44354 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-44354 | HTTPS FTP |
-Validation report
| Summary document |  emd_44354_validation.pdf.gz emd_44354_validation.pdf.gz | 1.3 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_44354_full_validation.pdf.gz emd_44354_full_validation.pdf.gz | 1.3 MB | Display | |
| Data in XML |  emd_44354_validation.xml.gz emd_44354_validation.xml.gz | 21.7 KB | Display | |
| Data in CIF |  emd_44354_validation.cif.gz emd_44354_validation.cif.gz | 28 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-44354 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-44354 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-44354 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-44354 | HTTPS FTP |
-Related structure data
| Related structure data |  9brcMC  9b8oC  9b8pC  9b8qC  9brbC  9brdC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_44354.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_44354.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.37333 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_44354_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_44354_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Synaptic Vesicle V-ATPase with synaptophysin and SidK, State 2
| Entire | Name: Synaptic Vesicle V-ATPase with synaptophysin and SidK, State 2 |
|---|---|
| Components |
|
-Supramolecule #1: Synaptic Vesicle V-ATPase with synaptophysin and SidK, State 2
| Supramolecule | Name: Synaptic Vesicle V-ATPase with synaptophysin and SidK, State 2 type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: TFS FALCON 4i (4k x 4k) / Average electron dose: 37.5 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.9000000000000001 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



















 Z
Z Y
Y X
X