登録情報 データベース : EMDB / ID : EMD-4274タイトル CryoEM structure of E.coli RNA polymerase paused elongation complex without RNA hairpin bound to NusA E. coli RNA polymerase paused elongation complex bound to NusA without density for RNA hairpin 複合体 : Cryo-EM structure of E. coli RNA polymerase paused elongation complex without RNA hairpin bound to NusA複合体 : RNA polymerase and NusAタンパク質・ペプチド : DNA-directed RNA polymerase subunit alphaタンパク質・ペプチド : DNA-directed RNA polymerase subunit betaタンパク質・ペプチド : DNA-directed RNA polymerase subunit beta'タンパク質・ペプチド : DNA-directed RNA polymerase subunit omega複合体 : Nucleic acidsDNA : DNA (30-MER)RNA : RNA (5'-R(P*GP*AP*UP*GP*UP*GP*UP*GP*CP*U)-3')DNA : DNA (39-MER)リガンド : MAGNESIUM IONリガンド : ZINC ION / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
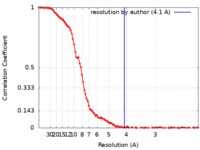
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Escherichia coli K-12 (大腸菌) / synthetic construct (人工物) / Escherichia coli (strain K12) (大腸菌)手法 / / 解像度 : 4.1 Å Guo X / Weixlbaumer A 資金援助 Organization Grant number 国 European Research Council ERC starting grant 679734
ジャーナル : Mol Cell / 年 : 2018タイトル : Structural Basis for NusA Stabilized Transcriptional Pausing.著者 : Xieyang Guo / Alexander G Myasnikov / James Chen / Corinne Crucifix / Gabor Papai / Maria Takacs / Patrick Schultz / Albert Weixlbaumer / 要旨 : Transcriptional pausing by RNA polymerases (RNAPs) is a key mechanism to regulate gene expression in all kingdoms of life and is a prerequisite for transcription termination. The essential bacterial ... Transcriptional pausing by RNA polymerases (RNAPs) is a key mechanism to regulate gene expression in all kingdoms of life and is a prerequisite for transcription termination. The essential bacterial transcription factor NusA stimulates both pausing and termination of transcription, thus playing a central role. Here, we report single-particle electron cryo-microscopy reconstructions of NusA bound to paused E. coli RNAP elongation complexes with and without a pause-enhancing hairpin in the RNA exit channel. The structures reveal four interactions between NusA and RNAP that suggest how NusA stimulates RNA folding, pausing, and termination. An asymmetric translocation intermediate of RNA and DNA converts the active site of the enzyme into an inactive state, providing a structural explanation for the inhibition of catalysis. Comparing RNAP at different stages of pausing provides insights on the dynamic nature of the process and the role of NusA as a regulatory factor. 履歴 登録 2018年1月26日 - ヘッダ(付随情報) 公開 2018年2月28日 - マップ公開 2018年3月7日 - 更新 2025年7月9日 - 現状 2025年7月9日 処理サイト : PDBe / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
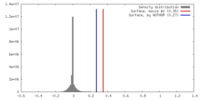
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報

 データ登録者
データ登録者 フランス, 1件
フランス, 1件  引用
引用 ジャーナル: Mol Cell / 年: 2018
ジャーナル: Mol Cell / 年: 2018

 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_4274.map.gz
emd_4274.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-4274-v30.xml
emd-4274-v30.xml emd-4274.xml
emd-4274.xml EMDBヘッダ
EMDBヘッダ emd_4274_fsc.xml
emd_4274_fsc.xml FSCデータファイル
FSCデータファイル emd_4274.png
emd_4274.png emd_4274_msk_1.map
emd_4274_msk_1.map マスクマップ
マスクマップ emd-4274.cif.gz
emd-4274.cif.gz emd_4274_half_map_1.map.gz
emd_4274_half_map_1.map.gz emd_4274_half_map_2.map.gz
emd_4274_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-4274
http://ftp.pdbj.org/pub/emdb/structures/EMD-4274 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4274
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4274 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_4274.map.gz / 形式: CCP4 / 大きさ: 83.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_4274.map.gz / 形式: CCP4 / 大きさ: 83.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_4274_msk_1.map
emd_4274_msk_1.map 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 画像解析
画像解析
 ムービー
ムービー コントローラー
コントローラー

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)