[English] 日本語
 Yorodumi
Yorodumi- EMDB-4118: CryoEM structure of the membrane pore complex of Pneumolysin at 4.5A -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4118 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of the membrane pore complex of Pneumolysin at 4.5A | |||||||||
 Map data Map data | PLY pore complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Bacterial toxins / pore complex / cryoEM structure / cholesterol-dependent cytolysin / Pneumolysin / membrane pore / TOXIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationcholesterol binding / toxin activity / killing of cells of another organism / host cell plasma membrane / extracellular region / membrane Similarity search - Function | |||||||||
| Biological species |   Streptococcus pneumoniae serotype 2 (strain D39 / NCTC 7466) (bacteria) Streptococcus pneumoniae serotype 2 (strain D39 / NCTC 7466) (bacteria) | |||||||||
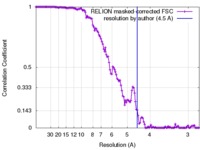
| Method | single particle reconstruction / cryo EM / Resolution: 4.5 Å | |||||||||
 Authors Authors | van Pee K / Neuhaus A | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: Elife / Year: 2017 Journal: Elife / Year: 2017Title: CryoEM structures of membrane pore and prepore complex reveal cytolytic mechanism of Pneumolysin. Authors: Katharina van Pee / Alexander Neuhaus / Edoardo D'Imprima / Deryck J Mills / Werner Kühlbrandt / Özkan Yildiz /  Abstract: Many pathogenic bacteria produce pore-forming toxins to attack and kill human cells. We have determined the 4.5 Å structure of the ~2.2 MDa pore complex of pneumolysin, the main virulence factor of ...Many pathogenic bacteria produce pore-forming toxins to attack and kill human cells. We have determined the 4.5 Å structure of the ~2.2 MDa pore complex of pneumolysin, the main virulence factor of , by cryoEM. The pneumolysin pore is a 400 Å ring of 42 membrane-inserted monomers. Domain 3 of the soluble toxin refolds into two ~85 Å β-hairpins that traverse the lipid bilayer and assemble into a 168-strand β-barrel. The pore complex is stabilized by salt bridges between β-hairpins of adjacent subunits and an internal α-barrel. The apolar outer barrel surface with large sidechains is immersed in the lipid bilayer, while the inner barrel surface is highly charged. Comparison of the cryoEM pore complex to the prepore structure obtained by electron cryo-tomography and the x-ray structure of the soluble form reveals the detailed mechanisms by which the toxin monomers insert into the lipid bilayer to perforate the target membrane. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4118.map.gz emd_4118.map.gz | 19.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4118-v30.xml emd-4118-v30.xml emd-4118.xml emd-4118.xml | 11.3 KB 11.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4118_fsc.xml emd_4118_fsc.xml | 12.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_4118.png emd_4118.png | 148.8 KB | ||
| Filedesc metadata |  emd-4118.cif.gz emd-4118.cif.gz | 5.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4118 http://ftp.pdbj.org/pub/emdb/structures/EMD-4118 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4118 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4118 | HTTPS FTP |
-Validation report
| Summary document |  emd_4118_validation.pdf.gz emd_4118_validation.pdf.gz | 265.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_4118_full_validation.pdf.gz emd_4118_full_validation.pdf.gz | 264.2 KB | Display | |
| Data in XML |  emd_4118_validation.xml.gz emd_4118_validation.xml.gz | 12.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4118 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4118 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4118 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4118 | HTTPS FTP |
-Related structure data
| Related structure data |  5ly6MC  5aoeC  5aofC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_4118.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4118.map.gz / Format: CCP4 / Size: 178 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | PLY pore complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.4 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Pneumolysin pore complex
| Entire | Name: Pneumolysin pore complex |
|---|---|
| Components |
|
-Supramolecule #1: Pneumolysin pore complex
| Supramolecule | Name: Pneumolysin pore complex / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 2.2 MDa |
-Macromolecule #1: Pneumolysin
| Macromolecule | Name: Pneumolysin / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Streptococcus pneumoniae serotype 2 (strain D39 / NCTC 7466) (bacteria) Streptococcus pneumoniae serotype 2 (strain D39 / NCTC 7466) (bacteria) |
| Molecular weight | Theoretical: 52.866066 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MANKAVNDFI LAMNYDKKKL LTHQGESIEN RFIKEGNQLP DEFVVIERKK RSLSTNTSDI SVTATNDSRL YPGALLVVDE TLLENNPTL LAVDRAPMTY SIDLPGLASS DSFLQVEDPS NSSVRGAVND LLAKWHQDYG QVNNVPARMQ YEKITAHSME Q LKVKFGSD ...String: MANKAVNDFI LAMNYDKKKL LTHQGESIEN RFIKEGNQLP DEFVVIERKK RSLSTNTSDI SVTATNDSRL YPGALLVVDE TLLENNPTL LAVDRAPMTY SIDLPGLASS DSFLQVEDPS NSSVRGAVND LLAKWHQDYG QVNNVPARMQ YEKITAHSME Q LKVKFGSD FEKAANSLDI DFNAVHSGEK QIQIVNFKQI YYTVSVDAVK NPGDVFQDTV TVEDLKQRGI SAERPLVYIS SV AYGRQVY LKLETTSKSD EVQAAFEAAI LGVKVAPQTQ WKQILDNTEV KAVILGGDPS SGARVVTGKV DMVEDLIQEG SRF TADHPG LPISYTTSFL RDNVVATFQN STDYVETKVT AYRNGDLLLD HSGAYVAQYY ITWDELSYDH QGKEVLTPKA WDRN GQDLT AHFTTSIPLK GNVRNLSVKI RECTGLAWEW WRTVYEKTDL PLVRKRTISI WGTTLYPQVE DKVEND UniProtKB: Pneumolysin |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL |
|---|---|
| Buffer | pH: 7 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average exposure time: 6.0 sec. / Average electron dose: 1.02 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)