[English] 日本語
 Yorodumi
Yorodumi- EMDB-36223: Cryo-EM structure of the GI.4 Chiba VLP complexed with the CV-1A1... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the GI.4 Chiba VLP complexed with the CV-1A1 Fv-clasp | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | NOROVIRUS / GI.4 / VIRUS LIKE PARTICLE / human monoclonal antibody / Fv-clasp / VIRUS | |||||||||
| Biological species |  Chiba virus / Chiba virus /  Homo sapiens (human) Homo sapiens (human) | |||||||||
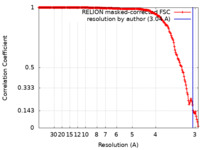
| Method | single particle reconstruction / cryo EM / Resolution: 3.04 Å | |||||||||
 Authors Authors | Hosaka T / Katsura K / Kimura-Someya T / Someya Y / Shirouzu M | |||||||||
| Funding support |  Japan, 2 items Japan, 2 items
| |||||||||
 Citation Citation |  Journal: J Virol / Year: 2024 Journal: J Virol / Year: 2024Title: Structural analyses of the GI.4 norovirus by cryo-electron microscopy and X-ray crystallography revealing binding sites for human monoclonal antibodies. Authors: Tomomi Kimura-Someya / Kazushige Katsura / Miyuki Kato-Murayama / Toshiaki Hosaka / Tomomi Uchikubo-Kamo / Kentaro Ihara / Kazuharu Hanada / Shin Sato / Kazutaka Murayama / Michiyo Kataoka / ...Authors: Tomomi Kimura-Someya / Kazushige Katsura / Miyuki Kato-Murayama / Toshiaki Hosaka / Tomomi Uchikubo-Kamo / Kentaro Ihara / Kazuharu Hanada / Shin Sato / Kazutaka Murayama / Michiyo Kataoka / Mikako Shirouzu / Yuichi Someya /  Abstract: Noroviruses are major causative agents of acute nonbacterial gastroenteritis in humans. There are neither antiviral therapeutic agents nor vaccines for noroviruses at this time. To evaluate the ...Noroviruses are major causative agents of acute nonbacterial gastroenteritis in humans. There are neither antiviral therapeutic agents nor vaccines for noroviruses at this time. To evaluate the potential usefulness of two previously isolated human monoclonal antibody fragments, CV-1A1 and CV-2F5, we first conducted a single-particle analysis to determine the cryo-electron microscopy structure of virus-like particles (VLPs) from the genogroup I genotype 4 (GI.4) Chiba strain uniformly coated with CV-1A1 fragments. The results revealed that the GI.4-specific CV-1A1 antibody bound to the P2 subdomain, in which amino acids are less conserved and variable. Interestingly, a part of the CV-1A1 intrudes into the histo-blood group antigen-binding site, suggesting that this antibody might exert neutralizing activity. Next, we determined the crystal structure of the protruding (P) domain of the capsid protein in the complex form with the CV-2F5 antibody fragment. Consistent with the cross-reactivity, the CV-2F5 bound to the P1 subdomain, which is rich in amino acids conserved among the GI strains, and moreover induced a disruption of Chiba VLPs. These results suggest that the broadly reactive CV-2F5 antibody can be used as both a universal detection reagent and an antiviral drug for GI noroviruses. IMPORTANCE: We conducted the structural analyses of the VP1 protein from the GI.4 Chiba norovirus to identify the binding sites of the previously isolated human monoclonal antibodies CV-1A1 and CV- ...IMPORTANCE: We conducted the structural analyses of the VP1 protein from the GI.4 Chiba norovirus to identify the binding sites of the previously isolated human monoclonal antibodies CV-1A1 and CV-2F5. The cryo-electron microscopy of the Chiba virus-like particles (VLPs) complexed with the Fv-clasp forms of GI.4-specific CV-1A1 revealed that this antibody binds to the highly variable P2 subdomain, suggesting that this antibody may have neutralizing ability against the GI.4 strains. X-ray crystallography revealed that the CV-2F5 antibody bound to the P1 subdomain, which is rich in conserved amino acids. This result is consistent with the ability of the CV-2F5 antibody to react with a wide variety of GI norovirus strains. It is also found that the CV-2F5 antibody caused a disruption of VLPs. Our findings, together with previous reports on the structures of VP1 proteins and VLPs, are expected to open a path for the structure-based development of antivirals and vaccines against norovirus disease. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_36223.map.gz emd_36223.map.gz | 449.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-36223-v30.xml emd-36223-v30.xml emd-36223.xml emd-36223.xml | 19.2 KB 19.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_36223_fsc.xml emd_36223_fsc.xml | 17.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_36223.png emd_36223.png | 191.9 KB | ||
| Masks |  emd_36223_msk_1.map emd_36223_msk_1.map | 476.8 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-36223.cif.gz emd-36223.cif.gz | 6.1 KB | ||
| Others |  emd_36223_additional_1.map.gz emd_36223_additional_1.map.gz emd_36223_half_map_1.map.gz emd_36223_half_map_1.map.gz emd_36223_half_map_2.map.gz emd_36223_half_map_2.map.gz | 13.2 MB 441.2 MB 441.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-36223 http://ftp.pdbj.org/pub/emdb/structures/EMD-36223 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36223 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-36223 | HTTPS FTP |
-Validation report
| Summary document |  emd_36223_validation.pdf.gz emd_36223_validation.pdf.gz | 936.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_36223_full_validation.pdf.gz emd_36223_full_validation.pdf.gz | 935.7 KB | Display | |
| Data in XML |  emd_36223_validation.xml.gz emd_36223_validation.xml.gz | 25.4 KB | Display | |
| Data in CIF |  emd_36223_validation.cif.gz emd_36223_validation.cif.gz | 34 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36223 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36223 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36223 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-36223 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_36223.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_36223.map.gz / Format: CCP4 / Size: 476.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.47 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_36223_msk_1.map emd_36223_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: #1
| File | emd_36223_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_36223_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_36223_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Viral protein 1 with its antibody fragments
| Entire | Name: Viral protein 1 with its antibody fragments |
|---|---|
| Components |
|
-Supramolecule #1: Viral protein 1 with its antibody fragments
| Supramolecule | Name: Viral protein 1 with its antibody fragments / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Chiba virus / Strain: GI/Human/Japan/Chiba 407/1987 Chiba virus / Strain: GI/Human/Japan/Chiba 407/1987 |
-Macromolecule #1: VP1
| Macromolecule | Name: VP1 / type: protein_or_peptide / ID: 1 / Details: GenBank ID AB042808 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Chiba virus / Strain: GI/Human/Japan/Chiba 407/1987 Chiba virus / Strain: GI/Human/Japan/Chiba 407/1987 |
| Molecular weight | Theoretical: 58.108332 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MASKDATPSA DGATGAGQLV PEVNTADPIP IDPVAGSSTA APVAGQVNLI DPWIINNFVQ APQGEFTISP NNTPGDVLFD LQLGPHLNP FLSHLSQMYN GWVGNMRVRV VLAGNAFTAG KVIICCVPPG FQSRTLSIAQ ATLFPHVIAD VRTLDPVEVP L EDVRNVLY ...String: MASKDATPSA DGATGAGQLV PEVNTADPIP IDPVAGSSTA APVAGQVNLI DPWIINNFVQ APQGEFTISP NNTPGDVLFD LQLGPHLNP FLSHLSQMYN GWVGNMRVRV VLAGNAFTAG KVIICCVPPG FQSRTLSIAQ ATLFPHVIAD VRTLDPVEVP L EDVRNVLY HNNDTQPTMR LLCMLYTPLR TGGASGGTDS FVVAGRVLTC PGPDFNFLFL VPPTVEQKTR PFTVPNIPLK YL SNSRIPN PIEGMSLSPD QTQNVQFQNG RCTIDGQPLG TTPVSVSQLC KFRGRITSGQ RVLNLTELDG SPFMAFAAPA PAG FPDLGS CDWHIEMSKI PNSSTQNNPI VTNSVKPNSQ QFVPHLSSIT LDENVSSGGD YIGTIQWTSP PSDSGGANTN FWKI PDYGS SLAEASQLAP AVYPPGFNEV IVYFMASIPG PNQSGSPNLV PCLLPQEYIT HFISEQAPIQ GEAALLHYVD PDTNR NLGE FKLYPGGYLT CVPNSSSTGP QQLPLDGVFV FASWVSRFYQ LKPVGTAGPA RGRLGVRR |
-Macromolecule #2: VH,SARAH
| Macromolecule | Name: VH,SARAH / type: protein_or_peptide / ID: 2 Details: VH-SARAH chimera of linker (residues (-6)-0), VH (residues 1-118), and SARAH (residues 119-171) Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 19.847254 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSSGSSGEVQ LVESGAEVKK PGASVKVSCK ASGYTFTSLY MHWVRQAPGQ GLEWMGMINP SGGGTWNAQK FQGRVTMTRD TSTSTVYME LRSLRSDDTA MYYCARDSDQ YSQGLGYWGQ GTLVTVCSGS DYEFLKSWTV EDLQKRLLAL DPMMEQEIEE I RQKYQSKR QPILDAIEAK |
-Macromolecule #3: VL,SARAH
| Macromolecule | Name: VL,SARAH / type: protein_or_peptide / ID: 3 Details: VL-SARAH chimera of linker (residues (-6)-0), VL (residues 1-112), and SARAH (residues 113-162) Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 18.19315 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GSSGSSGQSA LTQPASVSGS PGQSITISCT GTSSDVGGYN YVSWYQQHPG KAPKLMIYDV SKRPSGVSNR FSGSKSGNTA SLTISGLQA KDEADYYCSS YTSSSTWVFG GGTKLTVLGG SDYEFLKSWT VEDLQKRLLA LDPMMEQEIE EIRQKYQCKR Q PILDAIEA K |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.8 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




 Z
Z Y
Y X
X