+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of ACE2 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | receptor of SARS-CoV-2 / PROTEIN BINDING | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of amino acid transport / angiotensin-converting enzyme 2 / positive regulation of L-proline import across plasma membrane / Hydrolases; Acting on peptide bonds (peptidases); Metallocarboxypeptidases / angiotensin-mediated drinking behavior / regulation of systemic arterial blood pressure by renin-angiotensin / tryptophan transport / positive regulation of gap junction assembly / regulation of vasoconstriction / regulation of cardiac conduction ...positive regulation of amino acid transport / angiotensin-converting enzyme 2 / positive regulation of L-proline import across plasma membrane / Hydrolases; Acting on peptide bonds (peptidases); Metallocarboxypeptidases / angiotensin-mediated drinking behavior / regulation of systemic arterial blood pressure by renin-angiotensin / tryptophan transport / positive regulation of gap junction assembly / regulation of vasoconstriction / regulation of cardiac conduction / peptidyl-dipeptidase activity / maternal process involved in female pregnancy / angiotensin maturation / Metabolism of Angiotensinogen to Angiotensins / metallocarboxypeptidase activity / carboxypeptidase activity / negative regulation of signaling receptor activity / Attachment and Entry / positive regulation of cardiac muscle contraction / regulation of cytokine production / viral life cycle / blood vessel diameter maintenance / brush border membrane / regulation of transmembrane transporter activity / negative regulation of smooth muscle cell proliferation / negative regulation of ERK1 and ERK2 cascade / cilium / metallopeptidase activity / endocytic vesicle membrane / positive regulation of reactive oxygen species metabolic process / virus receptor activity / regulation of cell population proliferation / regulation of inflammatory response / endopeptidase activity / Induction of Cell-Cell Fusion / Potential therapeutics for SARS / entry receptor-mediated virion attachment to host cell / receptor-mediated endocytosis of virus by host cell / membrane fusion / Attachment and Entry / receptor-mediated virion attachment to host cell / symbiont entry into host cell / membrane raft / apical plasma membrane / endoplasmic reticulum lumen / cell surface / extracellular space / zinc ion binding / extracellular exosome / extracellular region / identical protein binding / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Xu J / Liu N / Wang HW | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Self-assembled monolayers guided free-standing atomic-crystal/molecule superstructure Authors: Zheng L / Xu J / Wang W / Gao X / Liu N / Wang HW / Peng HL | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_34979.map.gz emd_34979.map.gz | 7.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-34979-v30.xml emd-34979-v30.xml emd-34979.xml emd-34979.xml | 12.9 KB 12.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_34979.png emd_34979.png | 136.9 KB | ||
| Filedesc metadata |  emd-34979.cif.gz emd-34979.cif.gz | 5.3 KB | ||
| Others |  emd_34979_half_map_1.map.gz emd_34979_half_map_1.map.gz emd_34979_half_map_2.map.gz emd_34979_half_map_2.map.gz | 7.1 MB 7.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-34979 http://ftp.pdbj.org/pub/emdb/structures/EMD-34979 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34979 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-34979 | HTTPS FTP |
-Validation report
| Summary document |  emd_34979_validation.pdf.gz emd_34979_validation.pdf.gz | 772.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_34979_full_validation.pdf.gz emd_34979_full_validation.pdf.gz | 772.1 KB | Display | |
| Data in XML |  emd_34979_validation.xml.gz emd_34979_validation.xml.gz | 8.7 KB | Display | |
| Data in CIF |  emd_34979_validation.cif.gz emd_34979_validation.cif.gz | 10 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34979 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34979 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34979 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-34979 | HTTPS FTP |
-Related structure data
| Related structure data |  8hrnMC  8hriC  8hrjC  8hrkC  8hrlC  8hrmC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_34979.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_34979.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.0382 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_34979_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
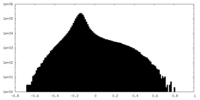
| Projections & Slices |
| ||||||||||||
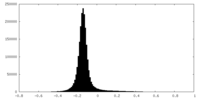
| Density Histograms |
-Half map: #1
| File | emd_34979_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
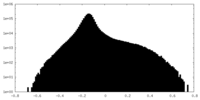
| Projections & Slices |
| ||||||||||||
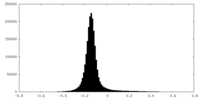
| Density Histograms |
- Sample components
Sample components
-Entire : ACE2
| Entire | Name: ACE2 |
|---|---|
| Components |
|
-Supramolecule #1: ACE2
| Supramolecule | Name: ACE2 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all / Details: on graphene/SAMs membranes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Angiotensin-converting enzyme 2
| Macromolecule | Name: Angiotensin-converting enzyme 2 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO / EC number: angiotensin-converting enzyme 2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 72.396492 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MHSSALLCCL VLLTGVRAQS TIEEQAKTFL DKFNHEAEDL FYQSSLASWN YNTNITEENV QNMNNAGDKW SAFLKEQSTL AQMYPLQEI QNLTVKLQLQ ALQQNGSSVL SEDKSKRLNT ILNTMSTIYS TGKVCNPDNP QECLLLEPGL NEIMANSLDY N ERLWAWES ...String: MHSSALLCCL VLLTGVRAQS TIEEQAKTFL DKFNHEAEDL FYQSSLASWN YNTNITEENV QNMNNAGDKW SAFLKEQSTL AQMYPLQEI QNLTVKLQLQ ALQQNGSSVL SEDKSKRLNT ILNTMSTIYS TGKVCNPDNP QECLLLEPGL NEIMANSLDY N ERLWAWES WRSEVGKQLR PLYEEYVVLK NEMARANHYE DYGDYWRGDY EVNGVDGYDY SRGQLIEDVE HTFEEIKPLY EH LHAYVRA KLMNAYPSYI SPIGCLPAHL LGDMWGRFWT NLYSLTVPFG QKPNIDVTDA MVDQAWDAQR IFKEAEKFFV SVG LPNMTQ GFWENSMLTD PGNVQKAVCH PTAWDLGKGD FRILMCTKVT MDDFLTAHHE MGHIQYDMAY AAQPFLLRNG ANEG FHEAV GEIMSLSAAT PKHLKSIGLL SPDFQEDNET EINFLLKQAL TIVGTLPFTY MLEKWRWMVF KGEIPKDQWM KKWWE MKRE IVGVVEPVPH DETYCDPASL FHVSNDYSFI RYYTRTLYQF QFQEALCQAA KHEGPLHKCD ISNSTEAGQK LFNMLR LGK SEPWTLALEN VVGAKNMNVR PLLNYFEPLF TWLKDQNKNS FVGWSTDWSP YADHHHHHHH HH UniProtKB: Angiotensin-converting enzyme 2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.5 µm / Nominal defocus min: 1.2 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 35987 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller











 Z
Z Y
Y X
X