+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-31519 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | structure of a channel | |||||||||
 Map data Map data | Cryo-EM map of a channel | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | ion channel / four-domain / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationcell communication involved in cardiac conduction / voltage-gated sodium channel activity involved in AV node cell action potential / voltage-gated sodium channel activity involved in bundle of His cell action potential / voltage-gated sodium channel activity involved in Purkinje myocyte action potential / voltage-gated sodium channel activity involved in SA node cell action potential / bundle of His cell action potential / regulation of ventricular cardiac muscle cell membrane depolarization / AV node cell action potential / SA node cell action potential / AV node cell to bundle of His cell communication ...cell communication involved in cardiac conduction / voltage-gated sodium channel activity involved in AV node cell action potential / voltage-gated sodium channel activity involved in bundle of His cell action potential / voltage-gated sodium channel activity involved in Purkinje myocyte action potential / voltage-gated sodium channel activity involved in SA node cell action potential / bundle of His cell action potential / regulation of ventricular cardiac muscle cell membrane depolarization / AV node cell action potential / SA node cell action potential / AV node cell to bundle of His cell communication / membrane depolarization during SA node cell action potential / response to denervation involved in regulation of muscle adaptation / sodium channel complex / membrane depolarization during atrial cardiac muscle cell action potential / cardiac ventricle development / voltage-gated sodium channel activity involved in cardiac muscle cell action potential / regulation of atrial cardiac muscle cell membrane repolarization / brainstem development / membrane depolarization during AV node cell action potential / regulation of atrial cardiac muscle cell membrane depolarization / membrane depolarization during bundle of His cell action potential / positive regulation of action potential / atrial cardiac muscle cell action potential / membrane depolarization during Purkinje myocyte cell action potential / membrane depolarization during cardiac muscle cell action potential / telencephalon development / membrane depolarization during action potential / positive regulation of sodium ion transport / regulation of sodium ion transmembrane transport / ventricular cardiac muscle cell action potential / regulation of ventricular cardiac muscle cell membrane repolarization / cardiac muscle cell action potential involved in contraction / voltage-gated sodium channel complex / sodium ion import across plasma membrane / regulation of cardiac muscle cell contraction / voltage-gated sodium channel activity / ankyrin binding / odontogenesis of dentin-containing tooth / sodium ion transport / regulation of heart rate by cardiac conduction / fibroblast growth factor binding / nitric-oxide synthase binding / intercalated disc / lateral plasma membrane / host cell membrane / membrane depolarization / positive regulation of heart rate / neuronal action potential / cardiac muscle contraction / T-tubule / sensory perception of pain / cellular response to calcium ion / regulation of heart rate / establishment of localization in cell / cerebellum development / positive regulation of epithelial cell proliferation / bioluminescence / sodium ion transmembrane transport / generation of precursor metabolites and energy / sarcolemma / caveola / Z disc / scaffold protein binding / transmembrane transporter binding / calmodulin binding / protein domain specific binding / axon / viral envelope / symbiont entry into host cell / ubiquitin protein ligase binding / protein kinase binding / virion attachment to host cell / nucleolus / perinuclear region of cytoplasm / virion membrane / enzyme binding / cell surface / endoplasmic reticulum / nucleoplasm / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |   Recombinant vesicular stomatitis Indiana virus rVSV-G/GFP Recombinant vesicular stomatitis Indiana virus rVSV-G/GFP | |||||||||
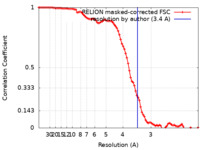
| Method | single particle reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Jiang DJ / Catterall WA | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Cell / Year: 2021 Journal: Cell / Year: 2021Title: Open-state structure and pore gating mechanism of the cardiac sodium channel. Authors: Daohua Jiang / Richard Banh / Tamer M Gamal El-Din / Lige Tonggu / Michael J Lenaeus / Régis Pomès / Ning Zheng / William A Catterall /    Abstract: The heartbeat is initiated by voltage-gated sodium channel Na1.5, which opens rapidly and triggers the cardiac action potential; however, the structural basis for pore opening remains unknown. Here, ...The heartbeat is initiated by voltage-gated sodium channel Na1.5, which opens rapidly and triggers the cardiac action potential; however, the structural basis for pore opening remains unknown. Here, we blocked fast inactivation with a mutation and captured the elusive open-state structure. The fast inactivation gate moves away from its receptor, allowing asymmetric opening of pore-lining S6 segments, which bend and rotate at their intracellular ends to dilate the activation gate to ∼10 Å diameter. Molecular dynamics analyses predict physiological rates of Na conductance. The open-state pore blocker propafenone binds in a high-affinity pose, and drug-access pathways are revealed through the open activation gate and fenestrations. Comparison with mutagenesis results provides a structural map of arrhythmia mutations that target the activation and fast inactivation gates. These results give atomic-level insights into molecular events that underlie generation of the action potential, open-state drug block, and fast inactivation of cardiac sodium channels, which initiate the heartbeat. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_31519.map.gz emd_31519.map.gz | 117.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-31519-v30.xml emd-31519-v30.xml emd-31519.xml emd-31519.xml | 16.8 KB 16.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_31519_fsc.xml emd_31519_fsc.xml | 9.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_31519.png emd_31519.png | 71 KB | ||
| Filedesc metadata |  emd-31519.cif.gz emd-31519.cif.gz | 7.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-31519 http://ftp.pdbj.org/pub/emdb/structures/EMD-31519 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31519 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-31519 | HTTPS FTP |
-Related structure data
| Related structure data |  7fbsMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_31519.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_31519.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM map of a channel | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.056 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : sodium channel with four homologous domains
| Entire | Name: sodium channel with four homologous domains |
|---|---|
| Components |
|
-Supramolecule #1: sodium channel with four homologous domains
| Supramolecule | Name: sodium channel with four homologous domains / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 250 KDa |
-Macromolecule #1: Sodium channel protein type 5 subunit alpha,Sodium channel protei...
| Macromolecule | Name: Sodium channel protein type 5 subunit alpha,Sodium channel protein type 5 subunit alpha,Sodium channel protein type 5 subunit alpha,G protein/GFP fusion protein type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Recombinant vesicular stomatitis Indiana virus rVSV-G/GFP Recombinant vesicular stomatitis Indiana virus rVSV-G/GFP |
| Molecular weight | Theoretical: 209.030625 KDa |
| Recombinant expression | Organism:  Mammalian 1 orthobornavirus Mammalian 1 orthobornavirus |
| Sequence | String: MANLLLPRGT SSFRRFTRES LAAIEKRMAE KQARGGSATS QESREGLQEE EAPRPQLDLQ ASKKLPDLYG NPPRELIGEP LEDLDPFYS TQKTFIVLNK GKTIFRFSAT NALYVLSPFH PVRRAAVKIL VHSLFSMLIM CTILTNCVFM AQHDPPPWTK Y VEYTFTAI ...String: MANLLLPRGT SSFRRFTRES LAAIEKRMAE KQARGGSATS QESREGLQEE EAPRPQLDLQ ASKKLPDLYG NPPRELIGEP LEDLDPFYS TQKTFIVLNK GKTIFRFSAT NALYVLSPFH PVRRAAVKIL VHSLFSMLIM CTILTNCVFM AQHDPPPWTK Y VEYTFTAI YTFESLVKIL ARGFCLHAFT FLRDPWNWLD FSVIVMAYTT EFVDLGNVSA LRTFRVLRAL KTISVISGLK TI VGALIQS VKKLADVMVL TVFCLSVFAL IGLQLFMGNL RHKCVRNFTE LNGTNGSVEA DGLVWNSLDV YLNDPANYLL KNG TTDVLL CGNSSDAGTC PEGYRCLKAG ENPDHGYTSF DSFAWAFLAL FRLMTQDCWE RLYQQTLRSA GKIYMIFFML VIFL GSFYL VNLILAVVAM AYEEQNQATI AETEEKEKRF QEAMEMLKKE HEALTIRGVD TVSRSSARQR ALSAVSVLTS ALEEL EESH RKCPPCWNRF AQHYLIWECC PLWMSIKQKV KFVVMDPFAD LTITMCIVLN TLFMALEHYN MTAEFEEMLQ VGNLVF TGI FTAEMTFKII ALDPYYYFQQ GWNIFDSIIV ILSLMELGLS RMGNLSVLRS FRLLRVFKLA KSWPTLNTLI KIIGNSV GA LGNLTLVLAI IVFIFAVVGM QLFGKNYSEL RHRISDSGLL PRWHMMDFFH AFLIIFRILC GEWIETMWDC MEVSGQSL C LLVFLLVMVI GNLVVLNLFL ALLLSSFSAD NLTAPDEDGE MNNLQLALAR IQRGLRFVKR TTWDFCCGIL RRRPKKPAA LATHSQLPSC ITAPRSPPPP EVEKVPPARK ETRFEEDKRP GQGTPGDSEP VCVPIAVAES DTEDQEEDEE NGKVWWRLRK TCYRIVEHS WFETFIIFMI LLSSGALAFE DIYLEERKTI KVLLEYADKM FTYVFVLEML LKWVAYGFKK YFTNAWCWLD F LIVDVSLV SLVANTLGFA EMGPIKSLRT LRALRPLRAL SRFEGMRVVV NALVGAIPSI MNVLLVCLIF WLIFSIMGVN LF AGKFGRC INQTEGDLPL NYTIVNNKSE CESFNVTGEL YWTKVKVNFD NVGAGYLALL QVATFKGWMD IMYAAVDSRG YEE QPQWED NLYMYIYFVV FIIFGSFFTL NLFIGVIIDN FNQQKKKLGG QDQQQTEEQK KYYNAMKKLG SKKPQKPIPR PLNK YQGFI FDIVTKQAFD VTIMFLICLN MVTMMVETDD QSPEKVNILA KINLLFVAIF TGECIVKMAA LRHYYFTNSW NIFDF VVVI LSIVGTVLSD IIQKYFFSPT LFRVIRLARI GRILRLIRGA KGIRTLLFAL MMSLPALFNI GLLLFLVMFI YSIFGM ANF AYVKWEAGID DMFNFQTFAN SMLCLFQITT SAGWDGLLSP ILNTGPPYCD PNLPNSNGSR GNCGSPAVGI LFFTTYI II SFLIVVNMYI AIILENFSVA TEESTEPLSE DDFDMFYEIW EKFDPEATQF IEYLALSDFA DALSEPLRIA KPNQISLI N MDLPMVSGDR IHCMDILFAF TKRVLGESGE MDALKIQMEE KFMAANPSKI SYEPITTTLE VLFQGPGSMV SKGEELFTG VVPILVELDG DVNGHKFSVS GEGEGDATYG KLTLKFICTT GKLPVPWPTL VTTLTYGVQC FSRYPDHMKQ HDFFKSAMPE GYVQERTIF FKDDGNYKTR AEVKFEGDTL VNRIELKGID FKEDGNILGH KLEYNYNSHN VYIMADKQKN GIKVNFKIRH N IEDGSVQL ADHYQQNTPI GDGPVLLPDN HYLSTQSALS KDPNEKRDHM VLLEFVTAAG ITLGMDELYK GSDYKDDDDK UniProtKB: Sodium channel protein type 5 subunit alpha, Sodium channel protein type 5 subunit alpha, Sodium channel protein type 5 subunit alpha, G protein/GFP fusion protein |
-Macromolecule #2: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 2 / Number of copies: 3 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #3: [(2~{R})-1-[2-azanylethoxy(oxidanyl)phosphoryl]oxy-3-hexadecanoyl...
| Macromolecule | Name: [(2~{R})-1-[2-azanylethoxy(oxidanyl)phosphoryl]oxy-3-hexadecanoyloxy-propan-2-yl] (~{Z})-octadec-9-enoate type: ligand / ID: 3 / Number of copies: 11 / Formula: 6OU |
|---|---|
| Molecular weight | Theoretical: 717.996 Da |
| Chemical component information |  ChemComp-6OU: |
-Macromolecule #4: 1-[2-[(2R)-2-oxidanyl-3-(propylamino)propoxy]phenyl]-3-phenyl-pro...
| Macromolecule | Name: 1-[2-[(2R)-2-oxidanyl-3-(propylamino)propoxy]phenyl]-3-phenyl-propan-1-one type: ligand / ID: 4 / Number of copies: 1 / Formula: 4Y4 |
|---|---|
| Molecular weight | Theoretical: 341.444 Da |
| Chemical component information | 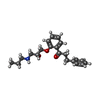 ChemComp-4Y4: |
-Macromolecule #5: (3beta,14beta,17beta,25R)-3-[4-methoxy-3-(methoxymethyl)butoxy]sp...
| Macromolecule | Name: (3beta,14beta,17beta,25R)-3-[4-methoxy-3-(methoxymethyl)butoxy]spirost-5-en type: ligand / ID: 5 / Number of copies: 2 / Formula: 9Z9 |
|---|---|
| Molecular weight | Theoretical: 544.805 Da |
| Chemical component information | 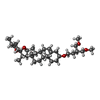 ChemComp-9Z9: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 5 mg/mL |
|---|---|
| Buffer | pH: 6 |
| Grid | Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum ER / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-7fbs: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)