[English] 日本語
 Yorodumi
Yorodumi- EMDB-29006: human EMC:human Cav1.2 channel complex in GDN detergent at 3.1 An... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | human EMC:human Cav1.2 channel complex in GDN detergent at 3.1 Angstrom | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | endoplasmic reticulum membrane protein complex / voltage-gated calcium channel / holdase / biogenesis / MEMBRANE PROTEIN | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Chen Z / Mondal A / Minor DL | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2023 Journal: Nat Struct Mol Biol / Year: 2023Title: Structural basis for Caαδ:gabapentin binding. Authors: Zhou Chen / Abhisek Mondal / Daniel L Minor /  Abstract: Gabapentinoid drugs for pain and anxiety act on the Caαδ-1 and Caαδ-2 subunits of high-voltage-activated calcium channels (Ca1s and Ca2s). Here we present the cryo-EM structure of the gabapentin- ...Gabapentinoid drugs for pain and anxiety act on the Caαδ-1 and Caαδ-2 subunits of high-voltage-activated calcium channels (Ca1s and Ca2s). Here we present the cryo-EM structure of the gabapentin-bound brain and cardiac Ca1.2/Caβ/Caαδ-1 channel. The data reveal a binding pocket in the Caαδ-1 dCache1 domain that completely encapsulates gabapentin and define Caαδ isoform sequence variations that explain the gabapentin binding selectivity of Caαδ-1 and Caαδ-2. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_29006.map.gz emd_29006.map.gz | 258.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-29006-v30.xml emd-29006-v30.xml emd-29006.xml emd-29006.xml | 16.9 KB 16.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_29006.png emd_29006.png | 49.3 KB | ||
| Others |  emd_29006_half_map_1.map.gz emd_29006_half_map_1.map.gz emd_29006_half_map_2.map.gz emd_29006_half_map_2.map.gz | 259.5 MB 259.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-29006 http://ftp.pdbj.org/pub/emdb/structures/EMD-29006 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29006 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-29006 | HTTPS FTP |
-Validation report
| Summary document |  emd_29006_validation.pdf.gz emd_29006_validation.pdf.gz | 920.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_29006_full_validation.pdf.gz emd_29006_full_validation.pdf.gz | 920 KB | Display | |
| Data in XML |  emd_29006_validation.xml.gz emd_29006_validation.xml.gz | 16.8 KB | Display | |
| Data in CIF |  emd_29006_validation.cif.gz emd_29006_validation.cif.gz | 20 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29006 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29006 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29006 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-29006 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_29006.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_29006.map.gz / Format: CCP4 / Size: 325 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 0.835 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_29006_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
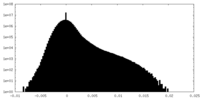
| Projections & Slices |
| ||||||||||||
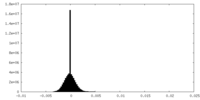
| Density Histograms |
-Half map: #2
| File | emd_29006_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
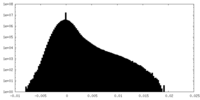
| Projections & Slices |
| ||||||||||||
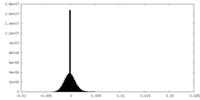
| Density Histograms |
- Sample components
Sample components
-Entire : Ternary complex of the human ER membrane protein complex (EMC) wi...
| Entire | Name: Ternary complex of the human ER membrane protein complex (EMC) with human CaV alpha1C and rabbit CaV beta3 |
|---|---|
| Components |
|
-Supramolecule #1: Ternary complex of the human ER membrane protein complex (EMC) wi...
| Supramolecule | Name: Ternary complex of the human ER membrane protein complex (EMC) with human CaV alpha1C and rabbit CaV beta3 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#11 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #2: Human CaV alpha1C
| Supramolecule | Name: Human CaV alpha1C / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #9 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 187 KDa |
-Supramolecule #3: ER membrane protein complex subunit 1
| Supramolecule | Name: ER membrane protein complex subunit 1 / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 112 KDa |
-Supramolecule #4: Rabbit CaV beta3
| Supramolecule | Name: Rabbit CaV beta3 / type: complex / ID: 4 / Parent: 1 / Macromolecule list: #10 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 100 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.7 mg/mL |
|---|---|
| Buffer | pH: 8 |
| Grid | Model: Quantifoil R1.2/1.3 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 46.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.7 µm / Nominal defocus min: 0.9 µm / Nominal magnification: 105000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: PDB ENTRY PDB model - PDB ID: Details: 6WW7 (PDB ID) and 7MIY (PDB ID) used to generate the startup model. |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 383185 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller







 Z
Z Y
Y X
X