+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of enolase from Streptococcus Pyogenes | |||||||||
 Map data Map data | Image of Cryo-Em Map of Octameric streptococcal Enolase | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | metalloenzyme / lyase / hPg-Receptor | |||||||||
| Function / homology |  Function and homology information Function and homology informationphosphopyruvate hydratase / phosphopyruvate hydratase complex / phosphopyruvate hydratase activity / peptidoglycan-based cell wall / glycolytic process / cell surface / magnesium ion binding / extracellular region Similarity search - Function | |||||||||
| Biological species |  Streptococcus pyogenes (bacteria) Streptococcus pyogenes (bacteria) | |||||||||
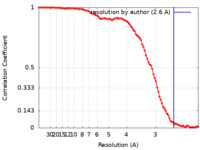
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Tjia-Fleck S / Readnour B / Castellino FJ | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Biochemistry / Year: 2023 Journal: Biochemistry / Year: 2023Title: High-Resolution Single-Particle Cryo-EM Hydrated Structure of Enolase Offers Insights into Its Function as a Plasminogen Receptor. Authors: Sheiny Tjia-Fleck / Bradley M Readnour / Yetunde A Ayinuola / Francis J Castellino /  Abstract: Cellular plasminogen (Pg) receptors (PgRs) are utilized to recruit Pg; stimulate its activation to the serine protease, plasmin (Pm); and sterically protect the surface Pm from inactivation by host ...Cellular plasminogen (Pg) receptors (PgRs) are utilized to recruit Pg; stimulate its activation to the serine protease, plasmin (Pm); and sterically protect the surface Pm from inactivation by host inhibitors. One such PgR is the moonlighting enzyme, enolase, some of which leaves the cytoplasm and resides at the cell surface to potentially function as a PgR. Since microbes employ conscription of host Pg by PgRs as one virulence mechanism, we explored the structural basis of the ability of enolase (Sen) to function in this manner. Employing single-particle cryo-electron microscopy (cryo-EM), recombinant Sen from was modeled at 2.6 Å as a stable symmetrical doughnut-shaped homooctamer with point group 422 (D4) symmetry, with a monomeric subunit molecular weight of ∼49 kDa. Binding sites for hPg were reported in other studies to include an internal K and the COOH-terminal K residues of Sen. However, in native Sen, the latter are buried within the minor interfaces of the octamer and do not function as a Pg-binding epitope. Whereas Sen and hPg do not interact in solution, when Sen is bound to a surface, hPg interacts with Sen independently of K. PgRs devoid of COOH-terminal lysine utilize lysine isosteres comprising a basic residue, "", and an anionic residue at " + 3" around one turn of an α-helix. We highlight a number of surface-exposed potential hPg-binding lysine isosteres and further conclude that while the octameric structure of Sen is critical for hPg binding, disruption of this octamer without dissociation exposes hPg-binding epitopes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26406.map.gz emd_26406.map.gz | 6.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26406-v30.xml emd-26406-v30.xml emd-26406.xml emd-26406.xml | 16.2 KB 16.2 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26406_fsc.xml emd_26406_fsc.xml | 11.6 KB | Display |  FSC data file FSC data file |
| Images |  emd_26406.png emd_26406.png | 133.5 KB | ||
| Masks |  emd_26406_msk_1.map emd_26406_msk_1.map | 64 MB |  Mask map Mask map | |
| Others |  emd_26406_half_map_1.map.gz emd_26406_half_map_1.map.gz emd_26406_half_map_2.map.gz emd_26406_half_map_2.map.gz | 6.3 MB 6.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26406 http://ftp.pdbj.org/pub/emdb/structures/EMD-26406 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26406 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26406 | HTTPS FTP |
-Validation report
| Summary document |  emd_26406_validation.pdf.gz emd_26406_validation.pdf.gz | 593.9 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_26406_full_validation.pdf.gz emd_26406_full_validation.pdf.gz | 593.5 KB | Display | |
| Data in XML |  emd_26406_validation.xml.gz emd_26406_validation.xml.gz | 16.3 KB | Display | |
| Data in CIF |  emd_26406_validation.cif.gz emd_26406_validation.cif.gz | 21.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26406 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26406 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26406 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26406 | HTTPS FTP |
-Related structure data
| Related structure data |  7uguMC  8dg4C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26406.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26406.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Image of Cryo-Em Map of Octameric streptococcal Enolase | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.29052 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_26406_msk_1.map emd_26406_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Cryo-Em Map 1 of Octameric streptococcal Enolase
| File | emd_26406_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Cryo-Em Map 1 of Octameric streptococcal Enolase | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half Cryo-Em Map 2 of Octameric streptococcal Enolase
| File | emd_26406_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Cryo-Em Map 2 of Octameric streptococcal Enolase | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Octameric structure of Enolase from Streoptococcus Pyogenes
| Entire | Name: Octameric structure of Enolase from Streoptococcus Pyogenes |
|---|---|
| Components |
|
-Supramolecule #1: Octameric structure of Enolase from Streoptococcus Pyogenes
| Supramolecule | Name: Octameric structure of Enolase from Streoptococcus Pyogenes type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Streptococcus pyogenes (bacteria) / Strain: AP53 Streptococcus pyogenes (bacteria) / Strain: AP53 |
| Molecular weight | Theoretical: 400 KDa |
-Macromolecule #1: Streptococcal Surface Enolase
| Macromolecule | Name: Streptococcal Surface Enolase / type: protein_or_peptide / ID: 1 / Enantiomer: DEXTRO |
|---|---|
| Source (natural) | Organism:  Streptococcus pyogenes (bacteria) / Strain: AP53 Streptococcus pyogenes (bacteria) / Strain: AP53 |
| Recombinant expression | Organism:  |
| Sequence | String: HMSIITDVYA REVLDSRGNP TLEVEVYTES GAFGRGMVPS GASTGEHEAV ELRDGDKSRY LGLGTQKAVD NVNNIIAEAI IGYDVRDQQA IDRAMIALDG TPNKGKLGAN AILGVSIAVA RAAADYLEVP LYTYLGGFNT KVLPTPMMNI INGGSHSDAP IAFQEFMIMP ...String: HMSIITDVYA REVLDSRGNP TLEVEVYTES GAFGRGMVPS GASTGEHEAV ELRDGDKSRY LGLGTQKAVD NVNNIIAEAI IGYDVRDQQA IDRAMIALDG TPNKGKLGAN AILGVSIAVA RAAADYLEVP LYTYLGGFNT KVLPTPMMNI INGGSHSDAP IAFQEFMIMP VGAPTFKEGL RWGAEVFHAL KKILKERGLV TAVGDEGGFA PKFEGTEDGV ETILKAIEAA GYEAGENGIM IGFDCASSEF YDKERKVYDY TKFEGEGAAV RTSAEQVDYL EELVNKYPII TIEDGMDEND WDGWKVLTER LGKRVQLVGD DFFVTNTEYL ARGIKENAAN SILIKVNQIG TLTETFEAIE MAKEAGYTAV VSHRSGETED STIADIAVAT NAGQIKTGSL SRTDRIAKYN QLLRIEDQLG EVAQYKGIKS FYNLKK |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL |
|---|---|
| Buffer | pH: 7.4 / Component - Concentration: 0.05 mM / Component - Formula: NaH2PO4 / Component - Name: Sodium phospate |
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Phase plate: VOLTA PHASE PLATE |
| Image recording | Film or detector model: GATAN ULTRASCAN 4000 (4k x 4k) / Number grids imaged: 1 / Number real images: 2756 / Average electron dose: 61.37 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 1.1 µm / Nominal magnification: 105000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller







 Z
Z Y
Y X
X