[English] 日本語
 Yorodumi
Yorodumi- EMDB-26037: E. coli 70S ribosomes bound with the ALS/FTD-associated dipeptide... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | E. coli 70S ribosomes bound with the ALS/FTD-associated dipeptide repeat protein PR20 | |||||||||||||||
 Map data Map data | E. coli 70S ribosome without tRNA and with peptidyl tunnel occupied by polyPR | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | ribosomal binding peptide / ALS/FTD-associated dipeptide repeat protein / RIBOSOME | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationDnaA-L2 complex / negative regulation of DNA-templated DNA replication initiation / ribosome assembly / assembly of large subunit precursor of preribosome / cytosolic ribosome assembly / ribosomal large subunit assembly / mRNA 5'-UTR binding / large ribosomal subunit / regulation of translation / ribosomal small subunit assembly ...DnaA-L2 complex / negative regulation of DNA-templated DNA replication initiation / ribosome assembly / assembly of large subunit precursor of preribosome / cytosolic ribosome assembly / ribosomal large subunit assembly / mRNA 5'-UTR binding / large ribosomal subunit / regulation of translation / ribosomal small subunit assembly / small ribosomal subunit / small ribosomal subunit rRNA binding / transferase activity / 5S rRNA binding / large ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / cytosolic large ribosomal subunit / tRNA binding / cytoplasmic translation / rRNA binding / negative regulation of translation / ribosome / structural constituent of ribosome / ribonucleoprotein complex / translation / response to antibiotic / mRNA binding / RNA binding / zinc ion binding / membrane / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||||||||
 Authors Authors | Loveland AB / Svidritskiy E / Susorov D / Lee S / Park A / Zvornicanin S / Demo G / Gao FB / Korostelev AA | |||||||||||||||
| Funding support |  United States, 4 items United States, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Ribosome inhibition by C9ORF72-ALS/FTD-associated poly-PR and poly-GR proteins revealed by cryo-EM. Authors: Anna B Loveland / Egor Svidritskiy / Denis Susorov / Soojin Lee / Alexander Park / Sarah Zvornicanin / Gabriel Demo / Fen-Biao Gao / Andrei A Korostelev /   Abstract: Toxic dipeptide-repeat (DPR) proteins are produced from expanded GC repeats in the C9ORF72 gene, the most common genetic cause of amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). ...Toxic dipeptide-repeat (DPR) proteins are produced from expanded GC repeats in the C9ORF72 gene, the most common genetic cause of amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). Two DPR proteins, poly-PR and poly-GR, repress cellular translation but the molecular mechanism remains unknown. Here we show that poly-PR and poly-GR of ≥20 repeats inhibit the ribosome's peptidyl-transferase activity at nanomolar concentrations, comparable to specific translation inhibitors. High-resolution cryogenic electron microscopy (cryo-EM) reveals that poly-PR and poly-GR block the polypeptide tunnel of the ribosome, extending into the peptidyl-transferase center (PTC). Consistent with these findings, the macrolide erythromycin, which binds in the tunnel, competes with poly-PR and restores peptidyl-transferase activity. Our results demonstrate that strong and specific binding of poly-PR and poly-GR in the ribosomal tunnel blocks translation, revealing the structural basis of their toxicity in C9ORF72-ALS/FTD. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_26037.map.gz emd_26037.map.gz | 283 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-26037-v30.xml emd-26037-v30.xml emd-26037.xml emd-26037.xml | 73.7 KB 73.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_26037_fsc.xml emd_26037_fsc.xml | 14.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_26037.png emd_26037.png | 44 KB | ||
| Filedesc metadata |  emd-26037.cif.gz emd-26037.cif.gz | 13.5 KB | ||
| Others |  emd_26037_additional_1.map.gz emd_26037_additional_1.map.gz emd_26037_half_map_1.map.gz emd_26037_half_map_1.map.gz emd_26037_half_map_2.map.gz emd_26037_half_map_2.map.gz | 284.3 MB 121.2 MB 121.2 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-26037 http://ftp.pdbj.org/pub/emdb/structures/EMD-26037 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26037 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-26037 | HTTPS FTP |
-Validation report
| Summary document |  emd_26037_validation.pdf.gz emd_26037_validation.pdf.gz | 880.4 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_26037_full_validation.pdf.gz emd_26037_full_validation.pdf.gz | 880 KB | Display | |
| Data in XML |  emd_26037_validation.xml.gz emd_26037_validation.xml.gz | 23.6 KB | Display | |
| Data in CIF |  emd_26037_validation.cif.gz emd_26037_validation.cif.gz | 30.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26037 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26037 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26037 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-26037 | HTTPS FTP |
-Related structure data
| Related structure data |  7tosMC  7tooC  7topC  7toqC  7torC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_26037.map.gz / Format: CCP4 / Size: 307.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_26037.map.gz / Format: CCP4 / Size: 307.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | E. coli 70S ribosome without tRNA and with peptidyl tunnel occupied by polyPR | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.87 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: E. coli 70S ribosome without tRNA and with...
| File | emd_26037_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | E. coli 70S ribosome without tRNA and with peptidyl tunnel occupied by polyPR, B-factor -35 applied | ||||||||||||
| Projections & Slices |
| ||||||||||||
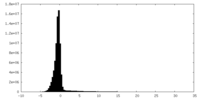
| Density Histograms |
-Half map: E. coli 70S ribosome without tRNA and with...
| File | emd_26037_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | E. coli 70S ribosome without tRNA and with peptidyl tunnel occupied by polyPR, half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
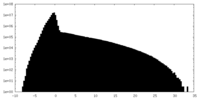
| Density Histograms |
-Half map: E. coli 70S ribosome without tRNA and withpeptidyl...
| File | emd_26037_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | E. coli 70S ribosome without tRNA and withpeptidyl tunnel occupied by polyPR, half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
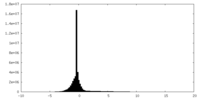
| Density Histograms |
- Sample components
Sample components
+Entire : E. coli 70S ribosome bound with the ALS/FTD-associated dipeptide ...
+Supramolecule #1: E. coli 70S ribosome bound with the ALS/FTD-associated dipeptide ...
+Macromolecule #1: 50S ribosomal protein L2
+Macromolecule #2: 50S ribosomal protein L3
+Macromolecule #3: 50S ribosomal protein L4
+Macromolecule #4: 50S ribosomal protein L5
+Macromolecule #5: 50S ribosomal protein L6
+Macromolecule #6: 50S ribosomal protein L9
+Macromolecule #7: 50S ribosomal protein L10
+Macromolecule #8: 50S ribosomal protein L11
+Macromolecule #9: 50S ribosomal protein L13
+Macromolecule #10: 50S ribosomal protein L14
+Macromolecule #11: 50S ribosomal protein L15
+Macromolecule #12: 50S ribosomal protein L16
+Macromolecule #13: 50S ribosomal protein L17
+Macromolecule #14: 50S ribosomal protein L18
+Macromolecule #15: 50S ribosomal protein L19
+Macromolecule #16: 50S ribosomal protein L20
+Macromolecule #17: Ribosomal protein L21
+Macromolecule #18: 50S ribosomal protein L22
+Macromolecule #19: 50S ribosomal protein L23
+Macromolecule #20: Ribosomal protein L24
+Macromolecule #21: 50S ribosomal protein L25
+Macromolecule #22: 50S ribosomal protein L27
+Macromolecule #23: 50S ribosomal protein L28
+Macromolecule #24: 50S ribosomal protein L29
+Macromolecule #25: 50S ribosomal protein L30
+Macromolecule #26: 50S ribosomal protein L31
+Macromolecule #27: 50S ribosomal protein L32
+Macromolecule #28: 50S ribosomal protein L33
+Macromolecule #29: 50S ribosomal protein L34
+Macromolecule #30: 50S ribosomal protein L35
+Macromolecule #31: 50S ribosomal protein L36
+Macromolecule #32: 30S ribosomal protein S2
+Macromolecule #33: 30S ribosomal protein S3
+Macromolecule #34: 30S ribosomal protein S4
+Macromolecule #35: 30S ribosomal protein S5
+Macromolecule #36: 30S ribosomal protein S6, non-modified isoform
+Macromolecule #37: 30S ribosomal protein S7
+Macromolecule #38: 30S ribosomal protein S8
+Macromolecule #39: 30S ribosomal protein S9
+Macromolecule #40: 30S ribosomal protein S10
+Macromolecule #41: 30S ribosomal protein S11
+Macromolecule #42: 30S ribosomal protein S12
+Macromolecule #43: 30S ribosomal protein S13
+Macromolecule #44: 30S ribosomal protein S14
+Macromolecule #45: 30S ribosomal protein S15
+Macromolecule #46: 30S ribosomal protein S16
+Macromolecule #47: 30S ribosomal protein S17
+Macromolecule #48: 30S ribosomal protein S18
+Macromolecule #49: 30S ribosomal protein S19
+Macromolecule #50: 30S ribosomal protein S20
+Macromolecule #51: Ribosomal protein S21
+Macromolecule #52: 50S ribosomal protein L1
+Macromolecule #56: PR20, ALS/FTD dipeptide repeat protein
+Macromolecule #53: 16S ribosomal RNA
+Macromolecule #54: 23S ribosomal RNA
+Macromolecule #55: 5S ribosomal RNA
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 35.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller













 Z
Z Y
Y X
X