+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-25481 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | 2.6 A structure of the nucleosome from a class without Chd1 | |||||||||
 Map data Map data | Sharpened 2.6A map in an orientation aligned to a common reference. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | CHD1 / chromatin remodeling / ATPase / DBD / nucleosome / remodeling / transcription / DNA BINDING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationstructural constituent of chromatin / nucleosome / heterochromatin formation / nucleosome assembly / protein heterodimerization activity / DNA binding / nucleus Similarity search - Function | |||||||||
| Biological species |  | |||||||||
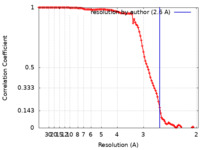
| Method | single particle reconstruction / cryo EM / Resolution: 2.6 Å | |||||||||
 Authors Authors | Nodelman IM / Das S / Faustino AM / Fried SD / Bowman GD / Armache J-P | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2022 Journal: Nat Struct Mol Biol / Year: 2022Title: Nucleosome recognition and DNA distortion by the Chd1 remodeler in a nucleotide-free state. Authors: Ilana M Nodelman / Sayan Das / Anneliese M Faustino / Stephen D Fried / Gregory D Bowman / Jean-Paul Armache /  Abstract: Chromatin remodelers are ATP-dependent enzymes that reorganize nucleosomes within all eukaryotic genomes. Here we report a complex of the Chd1 remodeler bound to a nucleosome in a nucleotide-free ...Chromatin remodelers are ATP-dependent enzymes that reorganize nucleosomes within all eukaryotic genomes. Here we report a complex of the Chd1 remodeler bound to a nucleosome in a nucleotide-free state, determined by cryo-EM to 2.3 Å resolution. The remodeler stimulates the nucleosome to absorb an additional nucleotide on each strand at two different locations: on the tracking strand within the ATPase binding site and on the guide strand one helical turn from the ATPase motor. Remarkably, the additional nucleotide on the tracking strand is associated with a local transformation toward an A-form geometry, explaining how sequential ratcheting of each DNA strand occurs. The structure also reveals a histone-binding motif, ChEx, which can block opposing remodelers on the nucleosome and may allow Chd1 to participate in histone reorganization during transcription. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25481.map.gz emd_25481.map.gz | 62 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25481-v30.xml emd-25481-v30.xml emd-25481.xml emd-25481.xml | 26.4 KB 26.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_25481_fsc.xml emd_25481_fsc.xml | 11.1 KB | Display |  FSC data file FSC data file |
| Images |  emd_25481.png emd_25481.png | 105 KB | ||
| Masks |  emd_25481_msk_1.map emd_25481_msk_1.map emd_25481_msk_2.map emd_25481_msk_2.map | 125 MB 125 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-25481.cif.gz emd-25481.cif.gz | 5.2 KB | ||
| Others |  emd_25481_additional_1.map.gz emd_25481_additional_1.map.gz emd_25481_additional_2.map.gz emd_25481_additional_2.map.gz emd_25481_additional_3.map.gz emd_25481_additional_3.map.gz emd_25481_additional_4.map.gz emd_25481_additional_4.map.gz emd_25481_half_map_1.map.gz emd_25481_half_map_1.map.gz emd_25481_half_map_2.map.gz emd_25481_half_map_2.map.gz | 33.6 MB 19.7 MB 64.4 MB 561.9 KB 115.9 MB 115.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25481 http://ftp.pdbj.org/pub/emdb/structures/EMD-25481 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25481 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25481 | HTTPS FTP |
-Related structure data
| Related structure data |  7swyFC  7tn2C C: citing same article ( F: fitted*YM |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10876 (Title: 2.3 A structure of the ATP-dependent chromatin remodeler Chd1 bound to the nucleosome in a nucleotide-free state EMPIAR-10876 (Title: 2.3 A structure of the ATP-dependent chromatin remodeler Chd1 bound to the nucleosome in a nucleotide-free stateData size: 4.7 TB Data #1: Unaligned frames of Chd1-bound nucleosomes collected on Gatan K3 in Super Resolution [micrographs - multiframe] Data #2: MotionCor2-aligned frames of Chd1-bound nucleosomes collected on Gatan K3 [micrographs - single frame] Data #3: Processed subsets [picked particles - single frame - processed]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_25481.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25481.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened 2.6A map in an orientation aligned to a common reference. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.972 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
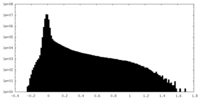
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_25481_msk_1.map emd_25481_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
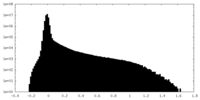
| Density Histograms |
-Mask #2
| File |  emd_25481_msk_2.map emd_25481_msk_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
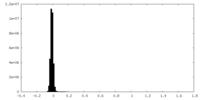
| Density Histograms |
-Additional map: Unsharpened 2.6A map in an orientation aligned to a common reference.
| File | emd_25481_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened 2.6A map in an orientation aligned to a common reference. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Unsharpened 2.6A map
| File | emd_25481_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened 2.6A map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Sharpened 2.6A map
| File | emd_25481_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened 2.6A map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: FSC Mask from cryoSPARC (before auto-tightening)
| File | emd_25481_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | FSC Mask from cryoSPARC (before auto-tightening) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half-map 1 (half A) from cryoSPARC
| File | emd_25481_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half-map 1 (half_A) from cryoSPARC | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: half-map 2 (half B) from cryoSPARC
| File | emd_25481_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half-map 2 (half_B) from cryoSPARC | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Chd1 ATP-dependent chromatin remodeler bound to a nucleosome in a...
| Entire | Name: Chd1 ATP-dependent chromatin remodeler bound to a nucleosome in a nucleotide-free state |
|---|---|
| Components |
|
-Supramolecule #1: Chd1 ATP-dependent chromatin remodeler bound to a nucleosome in a...
| Supramolecule | Name: Chd1 ATP-dependent chromatin remodeler bound to a nucleosome in a nucleotide-free state type: complex / ID: 1 / Parent: 0 / Details: This entry has a weak DNA-binding domain |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 400 KDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1.7 mg/mL |
|---|---|
| Buffer | pH: 7 Details: 20 mM HEPES, pH 7.0, 60 mM KCl, 1.5 mM DTT, 1 mM MgCl2 |
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
| Details | Nucleosome-bound CHD1, prepared in the presence of ATPgammaS, and then crosslinked using GraFix. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Digitization - Dimensions - Width: 11520 pixel / Digitization - Dimensions - Height: 8184 pixel / Number grids imaged: 1 / Number real images: 6906 / Average exposure time: 3.3 sec. / Average electron dose: 49.9 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated defocus max: 28.0 µm / Calibrated defocus min: 4.0 µm / Calibrated magnification: 46296 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: -2.2 µm / Nominal defocus min: -1.0 µm / Nominal magnification: 81000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)