[English] 日本語
 Yorodumi
Yorodumi- PDB-1gt8: DIHYDROPYRIMIDINE DEHYDROGENASE (DPD) FROM PIG, TERNARY COMPLEX W... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1gt8 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | DIHYDROPYRIMIDINE DEHYDROGENASE (DPD) FROM PIG, TERNARY COMPLEX WITH NADPH AND URACIL-4-ACETIC ACID | |||||||||
 Components Components | DIHYDROPYRIMIDINE DEHYDROGENASE | |||||||||
 Keywords Keywords | OXIDOREDUCTASE / ELECTRON TRANSFER / FLAVIN / IRON-SULFUR CLUSTERS / PYRIMIDINE CATABOLISM / 5-FLUOROURACIL DEGRADATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationdihydropyrimidine dehydrogenase (NADP+) / thymidine catabolic process / dihydropyrimidine dehydrogenase (NADP+) activity / uracil binding / beta-alanine biosynthetic process / thymine catabolic process / uracil catabolic process / FMN binding / NADP binding / flavin adenine dinucleotide binding ...dihydropyrimidine dehydrogenase (NADP+) / thymidine catabolic process / dihydropyrimidine dehydrogenase (NADP+) activity / uracil binding / beta-alanine biosynthetic process / thymine catabolic process / uracil catabolic process / FMN binding / NADP binding / flavin adenine dinucleotide binding / 4 iron, 4 sulfur cluster binding / protein homodimerization activity / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.3 Å MOLECULAR REPLACEMENT / Resolution: 3.3 Å | |||||||||
 Authors Authors | Dobritzsch, D. / Ricagno, S. / Schneider, G. / Schnackerz, K.D. / Lindqvist, Y. | |||||||||
 Citation Citation |  Journal: J. Biol. Chem. / Year: 2002 Journal: J. Biol. Chem. / Year: 2002Title: Crystal structure of the productive ternary complex of dihydropyrimidine dehydrogenase with NADPH and 5-iodouracil. Implications for mechanism of inhibition and electron transfer. Authors: Dobritzsch, D. / Ricagno, S. / Schneider, G. / Schnackerz, K.D. / Lindqvist, Y. #1: Journal: Acta Crystallogr.,Sect.D / Year: 2001 Title: Crystallization and Preliminary X-Ray Study of Pig Liver Dihydropyrimidine Dehydrogenase Authors: Dobritzsch, D. / Persson, K. / Schneider, G. / Lindqvist, Y. | |||||||||
| History |
| |||||||||
| Remark 700 | SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AF", "BF", "CF" AND "DF" IN EACH CHAIN ... SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AF", "BF", "CF" AND "DF" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 8-STRANDED BARREL, THIS IS REPRESENTED BY A 9-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1gt8.cif.gz 1gt8.cif.gz | 792.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1gt8.ent.gz pdb1gt8.ent.gz | 645.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1gt8.json.gz 1gt8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gt/1gt8 https://data.pdbj.org/pub/pdb/validation_reports/gt/1gt8 ftp://data.pdbj.org/pub/pdb/validation_reports/gt/1gt8 ftp://data.pdbj.org/pub/pdb/validation_reports/gt/1gt8 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||
| 2 | 
| ||||||||||||||||
| Unit cell |
| ||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS oper:
|
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 111603.344 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: Q28943, dihydropyrimidine dehydrogenase (NADP+) |
|---|
-Non-polymers , 5 types, 32 molecules 



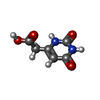




| #2: Chemical | ChemComp-SF4 / #3: Chemical | ChemComp-FMN / #4: Chemical | ChemComp-FAD / #5: Chemical | ChemComp-NDP / #6: Chemical | ChemComp-UAA / |
|---|
-Details
| Compound details | INVOLVED IN THE CATABOLISM OF URACIL AND THYMIDINE LEADING TO THE FORMATION OF BETA-ALANINE. ...INVOLVED IN THE CATABOLISM |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.45 Å3/Da / Density % sol: 40.03 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 7.5 Details: 100 MM SODIUM CITRATE PH 4.7, 16-20% POLYETHYLENE GLYCOL 6000, 1 MM DTT | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 293 K / pH: 4.7 / Method: vapor diffusion / Details: Dobritzsch, D., (2001) Acta Crystallogr., 57, 153. | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-3 / Wavelength: 0.931 / Beamline: ID14-3 / Wavelength: 0.931 |
| Detector | Type: MARRESEARCH / Detector: CCD / Date: Dec 12, 2000 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.931 Å / Relative weight: 1 |
| Reflection | Resolution: 3.3→30 Å / Num. obs: 62722 / % possible obs: 98.3 % / Redundancy: 3.5 % / Biso Wilson estimate: 28.1 Å2 / Rmerge(I) obs: 0.109 / Net I/σ(I): 9.8 |
| Reflection shell | Resolution: 3.3→3.36 Å / Redundancy: 3.2 % / Rmerge(I) obs: 0.463 / Mean I/σ(I) obs: 2.6 / % possible all: 92.9 |
| Reflection | *PLUS Highest resolution: 3.3 Å / Rmerge(I) obs: 0.098 |
| Reflection shell | *PLUS Highest resolution: 3.3 Å / % possible obs: 92.9 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: DIHYDROPYRIMIDINE DEHYDROGENASE, TERNARY COMPLEX WITH 5-IODOURACIL AND NADPH, PH 7.5 Resolution: 3.3→29.75 Å / Rfactor Rfree error: 0.008 / Data cutoff high absF: 2857472 / Isotropic thermal model: GROUP / Cross valid method: THROUGHOUT / σ(F): 0 Details: THE 6-8 C-TERMINAL RESIDUES AND A FEW LOOP-RESIDUES WERE NOT SEEN IN THE DENSITY DUE TO DISORDER
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 10 Å2 / ksol: 0.230877 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 53.3 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.3→29.75 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS | Rms dev Biso : 2 Å2 / Rms dev position: 0.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 3.3→3.45 Å / Rfactor Rfree error: 0.027 / Total num. of bins used: 8
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor obs: 0.217 / Rfactor Rfree: 0.273 / Rfactor Rwork: 0.217 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | *PLUS Rfactor obs: 0.294 |
 Movie
Movie Controller
Controller
















 PDBj
PDBj
















