+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Vertebrate microtubule-capping gamma-tubulin ring complex | |||||||||||||||||||||
 Map data Map data | Cryo-EM reconstruction of the vertebrate microtubule-capping gamma-tubulin ring complex isolated from Xenopus leavis egg extract | |||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||
 Keywords Keywords | cytoskeleton / microtubule / microtubule nucleation / complex / template / cap / gamma-tubulin / gamma-tubulin ring complex / CELL CYCLE | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationgamma-tubulin complex localization / equatorial microtubule organizing center / mitotic spindle microtubule / gamma-tubulin ring complex / microtubule minus-end binding / polar microtubule / gamma-tubulin complex / meiotic spindle organization / microtubule nucleation / gamma-tubulin binding ...gamma-tubulin complex localization / equatorial microtubule organizing center / mitotic spindle microtubule / gamma-tubulin ring complex / microtubule minus-end binding / polar microtubule / gamma-tubulin complex / meiotic spindle organization / microtubule nucleation / gamma-tubulin binding / mitotic sister chromatid segregation / spindle assembly / cytoplasmic microtubule organization / mitotic spindle organization / meiotic cell cycle / structural constituent of cytoskeleton / spindle pole / spindle / mitotic cell cycle / microtubule / centrosome / GTP binding / nucleus / cytoplasm Similarity search - Function | |||||||||||||||||||||
| Biological species | ||||||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 17.0 Å | |||||||||||||||||||||
 Authors Authors | Vermeulen BJA / Pfeffer S | |||||||||||||||||||||
| Funding support |  Germany, 6 items Germany, 6 items
| |||||||||||||||||||||
 Citation Citation |  Journal: EMBO J / Year: 2024 Journal: EMBO J / Year: 2024Title: γ-TuRC asymmetry induces local protofilament mismatch at the RanGTP-stimulated microtubule minus end. Authors: Bram Ja Vermeulen / Anna Böhler / Qi Gao / Annett Neuner / Erik Župa / Zhenzhen Chu / Martin Würtz / Ursula Jäkle / Oliver J Gruss / Stefan Pfeffer / Elmar Schiebel /   Abstract: The γ-tubulin ring complex (γ-TuRC) is a structural template for de novo microtubule assembly from α/β-tubulin units. The isolated vertebrate γ-TuRC assumes an asymmetric, open structure ...The γ-tubulin ring complex (γ-TuRC) is a structural template for de novo microtubule assembly from α/β-tubulin units. The isolated vertebrate γ-TuRC assumes an asymmetric, open structure deviating from microtubule geometry, suggesting that γ-TuRC closure may underlie regulation of microtubule nucleation. Here, we isolate native γ-TuRC-capped microtubules from Xenopus laevis egg extract nucleated through the RanGTP-induced pathway for spindle assembly and determine their cryo-EM structure. Intriguingly, the microtubule minus end-bound γ-TuRC is only partially closed and consequently, the emanating microtubule is locally misaligned with the γ-TuRC and asymmetric. In the partially closed conformation of the γ-TuRC, the actin-containing lumenal bridge is locally destabilised, suggesting lumenal bridge modulation in microtubule nucleation. The microtubule-binding protein CAMSAP2 specifically binds the minus end of γ-TuRC-capped microtubules, indicating that the asymmetric minus end structure may underlie recruitment of microtubule-modulating factors for γ-TuRC release. Collectively, we reveal a surprisingly asymmetric microtubule minus end protofilament organisation diverging from the regular microtubule structure, with direct implications for the kinetics and regulation of nucleation and subsequent modulation of microtubules during spindle assembly. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_19861.map.gz emd_19861.map.gz | 57.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-19861-v30.xml emd-19861-v30.xml emd-19861.xml emd-19861.xml | 28.3 KB 28.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_19861.png emd_19861.png | 104.8 KB | ||
| Filedesc metadata |  emd-19861.cif.gz emd-19861.cif.gz | 9.7 KB | ||
| Others |  emd_19861_half_map_1.map.gz emd_19861_half_map_1.map.gz emd_19861_half_map_2.map.gz emd_19861_half_map_2.map.gz | 49.5 MB 49.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-19861 http://ftp.pdbj.org/pub/emdb/structures/EMD-19861 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19861 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-19861 | HTTPS FTP |
-Validation report
| Summary document |  emd_19861_validation.pdf.gz emd_19861_validation.pdf.gz | 1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_19861_full_validation.pdf.gz emd_19861_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  emd_19861_validation.xml.gz emd_19861_validation.xml.gz | 12.2 KB | Display | |
| Data in CIF |  emd_19861_validation.cif.gz emd_19861_validation.cif.gz | 14.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19861 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19861 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19861 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-19861 | HTTPS FTP |
-Related structure data
| Related structure data |  9eojMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_19861.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_19861.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM reconstruction of the vertebrate microtubule-capping gamma-tubulin ring complex isolated from Xenopus leavis egg extract | ||||||||||||||||||||
| Voxel size | X=Y=Z: 2.004 Å | ||||||||||||||||||||
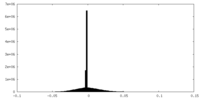
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: Half map 2 of the cryo-EM reconstruction of...
| File | emd_19861_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 of the cryo-EM reconstruction of the vertebrate microtubule-capping gamma-tubulin ring complex isolated from Xenopus leavis egg extract | ||||||||||||
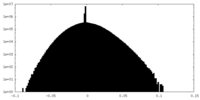
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map 1 of the cryo-EM reconstruction of...
| File | emd_19861_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 of the cryo-EM reconstruction of the vertebrate microtubule-capping gamma-tubulin ring complex isolated from Xenopus leavis egg extract | ||||||||||||
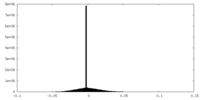
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Vertebrate microtubule-capping gamma-tubulin ring complex
| Entire | Name: Vertebrate microtubule-capping gamma-tubulin ring complex |
|---|---|
| Components |
|
-Supramolecule #1: Vertebrate microtubule-capping gamma-tubulin ring complex
| Supramolecule | Name: Vertebrate microtubule-capping gamma-tubulin ring complex type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Vertebrate microtubule-capping gamma-tubulin ring complex induced through the RanGTP spindle assembly pathway and isolated from Xenopus laevis egg extract |
|---|---|
| Source (natural) | Organism: |
-Macromolecule #1: Mitotic-spindle organizing protein 1
| Macromolecule | Name: Mitotic-spindle organizing protein 1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 7.749854 KDa |
| Sequence | String: MANASGNMSA VRETMDVLLE ISRLLNTGLD METLSICVRL CEQGINPEAL SSVIKELRRA SDTLKASEST AS UniProtKB: Mitotic-spindle organizing protein 1 |
-Macromolecule #2: Gamma-tubulin complex component 3 homolog
| Macromolecule | Name: Gamma-tubulin complex component 3 homolog / type: protein_or_peptide / ID: 2 / Number of copies: 5 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 103.789352 KDa |
| Sequence | String: MAVPDQKSPN VLLQNLCCRI LGKGEADVAQ QFQYAVRVIG SNFAPTVERD EFLVTEKIKK EFVRQRREAD GALFSELHRK LQSQGVLKN RWSILYLLLS LSEDPRKQPN KTSSFAALFA QALPRDAHST PYYYARPQSL PLSYQDRNVQ CAQNAASIGS S GISSIGMY ...String: MAVPDQKSPN VLLQNLCCRI LGKGEADVAQ QFQYAVRVIG SNFAPTVERD EFLVTEKIKK EFVRQRREAD GALFSELHRK LQSQGVLKN RWSILYLLLS LSEDPRKQPN KTSSFAALFA QALPRDAHST PYYYARPQSL PLSYQDRNVQ CAQNAASIGS S GISSIGMY ALNGPTPQSI IQGQSNQTPN MGDALRQQLG SRLAWTLAAG QQPSQQSTTT KGLPNTVSRN VPRTRREGDS SG SVEITET SLVRDLLYVF QGIDGKFVKM CNSENCYKVD GKVAVSKSLK DITSKLSELG WLHNKIKKYT DQRSLDRAFG LVG QSFCAA LHQELKEYYR LLSVLHSQLQ VEDDQGVNLG VESSLTLRRL LVWTFDPKIR LKTLAALVDH CQGRKGGELA SAVH AYTKT GDPYMRSLVQ HILGLVAYPI LNFLYRWIYD GELEDTYHEF FVASDPVVKT DRLWHDKYSL RKSMIPSFMT MDQSR KVLL IGKSINFLHQ VCHDQTPASK AMAVGKSAES PKDAAELFTD LENAFQTKID AAYFDTSKYL LDVLNKNYNL LEHMQA MRR YLLLGQGDFI RHLMDLLKPE LVRPATTLYQ HNLTGILETA VRATNAQFDN PEILKRLDVR LLEVSPGDTG WDVFSLD YH VDGPIATVFT RECMSHYLRV FNFLWRAKRM EYILTDIWKG HMCNAKLLKG MPELSGVLHQ CHILASEMVH FIHQMQYY I TFEVLECSWD ELWNKVLKAQ DLDHIIAAHD VFLDTIISRC LLDSESRALL NQLRAVFDQI IEFQNAQDAL YRAALEELQ QRLQFEERKK ERESEGEWGV TAAEEDVENK RIQEFQESIP KMRSQLRILT HFYQGIVQQF LVLLTTSTDE SLRFLSFRLD FNEHYKARE PRLRVSMGTR GRRSFHV UniProtKB: Gamma-tubulin complex component 3 homolog |
-Macromolecule #3: Gamma-tubulin complex component
| Macromolecule | Name: Gamma-tubulin complex component / type: protein_or_peptide / ID: 3 / Number of copies: 5 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 103.468312 KDa |
| Sequence | String: MSEFRIHHDV NELISLLHVF GLEGADVYID LLQKNRTPYV TTSVSTHSAK VKIAEFSRTP DDFLKKYEEL KSKNTRNLDP LVYLLSKLI EDKETLQYLQ QNAKDKAELA TSSVTSVSLP IAPNTSKISM QELEELRRQL ETATVAVSCS HQPVEVLRKF L RDKLNKKH ...String: MSEFRIHHDV NELISLLHVF GLEGADVYID LLQKNRTPYV TTSVSTHSAK VKIAEFSRTP DDFLKKYEEL KSKNTRNLDP LVYLLSKLI EDKETLQYLQ QNAKDKAELA TSSVTSVSLP IAPNTSKISM QELEELRRQL ETATVAVSCS HQPVEVLRKF L RDKLNKKH TGHPVPVFPS WVYERPALTG DFMSFSNPST DVTVSIGTLP LPSQETCLVE DLLYILIGVD GRYISVQPLV GR QSRSFSV EQNLDSSVKE LVNRILPVAT NYSTVTRFVE ENSSFEYGQV NHALGAAMRT LGKEYMILIS QLEHLQRQGL LSL QKLWFY IQPTLRTMEV LASIATSLNK GECFGGATLS LLHDRTFGYT GDSQAQELCL YLTKAASAPY FDILERWIYR GIIN DPYSE FMVEEHELQK EKIQEDYNDK YWDQRYTIVQ QQIPSFLQKV ADKILSTGKY LNVVRECGHD VTCPDAKEIT YTLKE QAYV ERIEKAYNYA SKVLLDFLME EEELVAHLRS IKHYFLMDQG DFFVHFMDLT EEELKKPVDD IIPTRLEALL ELALRM STA NTDPFKDDLK IELMPHDLIT QLLRVLAIET HQEKALINSD PTELALSGLE SFSFDYIVKW PLSLIINRKA LTRYQML FR HMFYCKHVER LLCNVWISNK TAKQFSLHSA KWFAGAFTLR QRMLNFVQNI QYYMMFEVME PTWHILEKNL KSASNIDD V LSHHTSFLDN CLKDCMLTNP ELLKIFSKLM SVCVMFTNCL QRFTQSMQVQ TEMEHLTLEH GTMMGPPTQC ERTEEALKK KLTSKYLEEH IDKFPSSFGF ESTINNFDSN FSAHLMDLLD KLSMYSTSDC EHSMINIIYR LDFNGFYTER LKQLSSERNQ KSAPLLGPA QHAVSTK UniProtKB: Gamma-tubulin complex component |
-Macromolecule #4: Gamma-tubulin complex component 6
| Macromolecule | Name: Gamma-tubulin complex component 6 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 193.069141 KDa |
| Sequence | String: MDSITKLFGD LCESHMVGFP WRTALNSRKH SKNRTKQTLK KLAYDTLFVH LFQDEARKLQ PNCTRLPVKN KIIMLSFNLR ICGMSSEAD RLEELVEYLE QSNGIQISDL HAVLELLVEL SGTGPPQLLP PKRDYFKNNK YVGRNVKYQG YDYYDVQVFE A DLGTTVAY ...String: MDSITKLFGD LCESHMVGFP WRTALNSRKH SKNRTKQTLK KLAYDTLFVH LFQDEARKLQ PNCTRLPVKN KIIMLSFNLR ICGMSSEAD RLEELVEYLE QSNGIQISDL HAVLELLVEL SGTGPPQLLP PKRDYFKNNK YVGRNVKYQG YDYYDVQVFE A DLGTTVAY QELEISTTIQ RTLQIMEAAP GTGLPALSFF SQNDLSTDKF EKETRGSLFG ALVHSRTNDM DIKLDMPPVP EN ADLSGLA IKVPQSIDQS EDEGFQSASN MTPDSQSEPS MTPDIDVWEA VLTYGPSKRR CWERIGCPPG KREEPYVTEA GRE AFDKLY KLHEGGLQIL SATTLQPQLV LLEETDLVKA VLNVLIGVVS STFSYNQALQ SFAVKQGVYI SGTSPDNVSS LLTQ VAEYG TYYTRLSHFS LLTVLDSSHS NGLVFQAFTS GLRKYLQYYR ACVLSTPASL TLLTISFLFR KLGRQLRYLA ELCCI GTLV TSATRGISTA FPTGVKLLSY LYKEALENSS NENYPVLLSL LKTSCEPYTR FIYDWVYSGV FRDVCGEFMI QVNEDY LGF RDKRYWTHGY VLISKEVEDC VPVFLKHVAN EIYICGKTIN LLKLCCPKHY ICWSDIPVPR ISVTFSLEEL KEMEKDC AV YVARMERIAR HSCISKEQKA LQTEIARQEL IIQARETTEK VFETFKDRKL AEKLSLDTKK RELFQKLKDQ YEKEQERR L TTKQEEADDD FSYAREIRDR EKRLKALEEE LELKTRQELI EHYSRLSEEA TRKEQRALWK LQRHKLETIR LKFFLEEQK RMQDLVANFP VDICEENLGV LPDGEISHQT DNTNDAGLGN IENEKSVPEQ HALHNNNDEV YTAQNCISKS ESLCVDVTLP TENVHSQTS NASVLGVPSF DSNLCTPDVD IIDFLPTLPS ENQEVAVVQS LVDDALISIG SDLNTDTKDK ESLCALKSDL Q ESSTGSEY DFKTILKPIA CTQVSQGHIK IGEYSSNVQP ARPRWSTHGH SSDSNIKIGN YVSDINVHQP KHSQHGHSSD SN INISDHM SDVEPRLPRL NLHGHISTGH IKVGEYASDV EPSTPRHSVH GHASQGNIKI GENVSDVKLS RPRWNIHGHV SDA NIKIGE NTSEIAPLRP RWNIHGHASQ SHIKIGELVS DIEPSQPRRT PFGHPSQSSI PIGDQPVEKY AQKSESEVHS SNST IQHLL YSNIPDKNKD TGGTLTDSPV PVPDQGNSND DTEKRSSTLE QRVQAADSVC DGEASPNTAQ SLPCMSDTLD FGTNG EENV GNDDHTWEKQ QEYLKGLAEK YCLEKYQDSY ELMSHPPVLH LYSNVMPNRF SFPTDSDIKS ATDETTVQLI ELLSLP VLM KYSVTAPMVS HVYLVNKAIV DYYFVELKME RHFEAMRHFL LMEDGEFAQS LSDMLFEKLG SGQTPSELLN PLVLNSI LN KALQYSLHGD SSLASNLTFA LKYLPEVFTP TAPDALSCLE LKYKVDWPLN IVITDTCMNK YSRIFSFLLQ LKHMVWTL R DVWFHLKRTA LVNQASNSVQ YRQLQLYRHE MQHFVKVIQG YIANQILHVT WCEFRNKLSA VSNLEEIYKT HADYLNKAL FRGLLTEKAA PLMNIIHSIF SLILKFRLQL ISQSWICDTG KQMAVHPNFG LMQQSYNTFK YYSDFLFEVV SKLVNRGYQP HLEDFLLRI NFNSYYKQS UniProtKB: Gamma-tubulin complex component 6 |
-Macromolecule #5: Tubulin gamma-1 chain
| Macromolecule | Name: Tubulin gamma-1 chain / type: protein_or_peptide / ID: 5 / Number of copies: 14 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 51.226719 KDa |
| Sequence | String: MPREIITLQL GQCGNQIGFE FWKQLCAEHG ISPEGIVEEF ATEGTDRKDV FFYQADDEHY IPRAVLLDLE PRVIHSILNS PYANLYNPE NIYLSEHGGG AGNNWASGFS QGEKIHEDIF DIIDREADGS DSLEGFVLCH SIAGGTGSGL GSYLLERLND R YPKKLVQT ...String: MPREIITLQL GQCGNQIGFE FWKQLCAEHG ISPEGIVEEF ATEGTDRKDV FFYQADDEHY IPRAVLLDLE PRVIHSILNS PYANLYNPE NIYLSEHGGG AGNNWASGFS QGEKIHEDIF DIIDREADGS DSLEGFVLCH SIAGGTGSGL GSYLLERLND R YPKKLVQT YSVFPNQDEM SHVVVQPYNS LLTLKRLTQN ADCVVVLDNT ALNRIATDRL HIQNPSFSQI NQLVSTIMSA ST TTLRYPG YMNNDLIGLI ASLIPTPRLH FLMTGYTPLT TDQSVASVRK TTVLDVMRRL LQPKNVMVST GRDRQTNHCY IAI LNIIQG EVDPTQVHKS LQRIRERKLA NFIPWGPASI QVALSRKSPY LPSAHRVSGL MMANHTNISS LFERTCRQYD KLRK REAFL EQFRKEDIFK DNFDELDNSR EIVQQLIDEY HAATRPDYIS WGTQDK UniProtKB: Tubulin gamma-1 chain |
-Macromolecule #6: Gamma-tubulin complex component
| Macromolecule | Name: Gamma-tubulin complex component / type: protein_or_peptide / ID: 6 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 117.577633 KDa |
| Sequence | String: MAHWTRFERD QEGDIKKLVS LMSGIQDDQD GNFQQALQFA WSNFRFHRYL DVSSHTVLRT LEGIFEKLVV HSDLEKAESW KRLTEEFLL LPLPNTEGTK TDSHFAVLSL LLCLSDSPSN HDYTEKPRKK ENDEQEPFDW GKYLREGEDI EFSPDADTPE W SEASEEED ...String: MAHWTRFERD QEGDIKKLVS LMSGIQDDQD GNFQQALQFA WSNFRFHRYL DVSSHTVLRT LEGIFEKLVV HSDLEKAESW KRLTEEFLL LPLPNTEGTK TDSHFAVLSL LLCLSDSPSN HDYTEKPRKK ENDEQEPFDW GKYLREGEDI EFSPDADTPE W SEASEEED AQEPPSREDS GIQVDRTPLE DPEKKGAPPL VSWKVGEPDA RSWLEQHIVH QYWTSRAPRF SHSSHLHSNL SA IWDQHLY TTDPLYTPDD KTIVTETQVI RETLWLLSGV KKLLIFQLND GKVNVRNDII VTHMTQNCLR SVLEQIAAYG QVV FRLQKF IDEITGHGSE VPLPGTLPTA KKTTEAPFRT YQAFMWALYK YFISFKEELT EIEKCIINKD ETVTLAIVLD KLAP RLAQL KVLHRVFSTG IAEVPPDTRN VVRASHLLNT LYKAILDYDN VGEASEQTVS LLFCLWVETV RPYLEIVDEW IVHGN LFDP AKEFIIQRNK DVPFNHRDFW YATYTLYSVS EKTENEDKMS DNASASSGSD QAPAGRQHTM VSFLKPVLKQ IIMAGK SMQ LLKNLKCRTA LQQDSSRDSD RKSLYTLFLE SVQSRLQHGN DSVPDIITEQ QVNKLSLIKM QSIVAKHLEL DEVHDPL LA INFVRLYLEQ SDFLETFTCN EVCVDRSSES VTCQSFELTL RSCLYPHIGK QYLECCGNLM YTLKKDYRLV EYLQAMRN F FLLEAGDTMY DFYTPIFDKI REKEPWLNLS YLNVQIQEAV GQRYPDDSTR LSVSFESVDL AKKKLPVHTL DGLILSYKV PWPVDIVISS ECQKIYNQVF LLLLLIKWAK YSLDVLQFNE LGNASENEST KEGATVEPFP LPPLTSPSEP KGQQIHRMFL LRVKLMHFV NSLHNYLMTR ILHSTGLEFQ HQVEEAKDLD QLIKIHYRYL STIHDRCLLR EKVSSVKEAI MKVLNVVLMF A DRWHAGLG AWKKESIVKM ESDFTNCHKF LVKVLNKAVC RGSFPHLESL ALSLMAGMEQ S UniProtKB: Gamma-tubulin complex component |
-Macromolecule #7: Gamma-tubulin complex component
| Macromolecule | Name: Gamma-tubulin complex component / type: protein_or_peptide / ID: 7 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: |
| Molecular weight | Theoretical: 76.122961 KDa |
| Sequence | String: MIHELLLALS GYPGSIFTWN KRTGLQVSQD IPFLHPGETS VLNRLCKLGT DYIRFTEFIE QYTGHVQQQD HHPSQQGQVG LHGIYLRAF CRGLDSILQP YRQALLDLEQ EFLADPHLSI SHINYSLDQF HLLFPSIMVV VEQIKSQKIH GCQILETVYK H SCGGLPPV ...String: MIHELLLALS GYPGSIFTWN KRTGLQVSQD IPFLHPGETS VLNRLCKLGT DYIRFTEFIE QYTGHVQQQD HHPSQQGQVG LHGIYLRAF CRGLDSILQP YRQALLDLEQ EFLADPHLSI SHINYSLDQF HLLFPSIMVV VEQIKSQKIH GCQILETVYK H SCGGLPPV RSALEKTLAV CHGVMYKQLS AWMLHGLLLD QYEEFFVRQG SSSGNLAAAF EEEEDDLGIG GLTGKQLREL QD LRLIEEE NMLAPSLKQF SLRAEMLPSY IPVRVAEKIL FVGESVQMFE NQNVNMSRTG SILKNQEDTF AAELHRLKQQ PLF SLVDFE SVLDRIRSTV AEHLWKLMVE ESDLLGQLKI IKDFYLLGRG ELFQAFIDVA QNMLKTPPTA VTEHDVNVAF QLSA HKVLL DDDNLLPLLN LTIDYHGKEH KDTSQPREGP FRDMSPREAP TSGWAALGLS YKVQWPLHIL FTPAVLEKYN VVFKY LLSV RRVQSELQHC WALQMQRKHL ESNKTDAIKW RLQNHMAFLV DNLQYYLQVD VLESQFSQLL QQINSTRDFE SIRLAH DHF LSNLLAQSFI LLKPVFHCLN EILELCHSFC SLVSQNLGPL DERGAGQLDI LVKGFSCQSS LLFRILSSVR NHQINPD LA QLLLRLDYNK YYTQAGGTLG SFGL UniProtKB: Gamma-tubulin complex component |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 6.8 |
|---|---|
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 43.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 33000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP EMDB ID: Details: GRIP2 and gamma-tubulin in spokes 5 and 6 were removed |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 17.0 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3.1) / Number images used: 8497 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.1) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.1) |
 Movie
Movie Controller
Controller







 Z
Z Y
Y X
X