[English] 日本語
 Yorodumi
Yorodumi- EMDB-17421: CryoEM structure of a GroEL7-GroES7 cage with encapsulated ordere... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
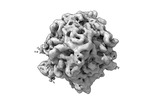
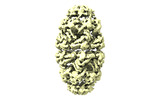
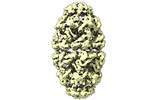
| Title | CryoEM structure of a GroEL7-GroES7 cage with encapsulated ordered substrate MetK in the presence of ADP-BeFx | |||||||||
 Map data Map data | GroEL7-GroES7 cage with encapsulated ordered substrate MetK in presence of ADP-BeFx | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Chaperonin / Folding cage / proteostasis / heat shock / ATPase / CHAPERONE | |||||||||
| Function / homology |  Function and homology information Function and homology informationmethionine adenosyltransferase / methionine adenosyltransferase activity / S-adenosylmethionine cycle / S-adenosylmethionine biosynthetic process / GroEL-GroES complex / chaperonin ATPase / mitochondrial unfolded protein response / protein import into mitochondrial intermembrane space / virion assembly / potassium ion binding ...methionine adenosyltransferase / methionine adenosyltransferase activity / S-adenosylmethionine cycle / S-adenosylmethionine biosynthetic process / GroEL-GroES complex / chaperonin ATPase / mitochondrial unfolded protein response / protein import into mitochondrial intermembrane space / virion assembly / potassium ion binding / chaperone cofactor-dependent protein refolding / positive regulation of interferon-alpha production / protein folding chaperone / isomerase activity / ATP-dependent protein folding chaperone / response to radiation / positive regulation of interleukin-6 production / positive regulation of type II interferon production / unfolded protein binding / positive regulation of T cell activation / protein folding / one-carbon metabolic process / protein-folding chaperone binding / response to heat / protein refolding / magnesium ion binding / ATP hydrolysis activity / ATP binding / identical protein binding / membrane / metal ion binding / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
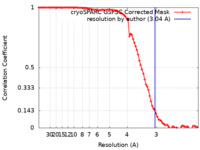
| Method | single particle reconstruction / cryo EM / Resolution: 3.04 Å | |||||||||
 Authors Authors | Wagner J / Beck F / Bracher A / Caravajal AI / Wan W / Bohn S / Koerner R / Baumeister W / Fernandez-Busnadiego R / Hartl FU | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Visualizing chaperonin function in situ by cryo-electron tomography Authors: Wagner J / Caravajal AI / Beck F / Bracher A / Wan W / Bohn S / Koerner R / Baumeister W / Fernandez-Busnadiego R / Hartl FU | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17421.map.gz emd_17421.map.gz | 59.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17421-v30.xml emd-17421-v30.xml emd-17421.xml emd-17421.xml | 25.7 KB 25.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17421_fsc.xml emd_17421_fsc.xml | 8.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_17421.png emd_17421.png | 101.9 KB | ||
| Filedesc metadata |  emd-17421.cif.gz emd-17421.cif.gz | 7.1 KB | ||
| Others |  emd_17421_half_map_1.map.gz emd_17421_half_map_1.map.gz emd_17421_half_map_2.map.gz emd_17421_half_map_2.map.gz | 59.4 MB 59.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17421 http://ftp.pdbj.org/pub/emdb/structures/EMD-17421 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17421 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17421 | HTTPS FTP |
-Validation report
| Summary document |  emd_17421_validation.pdf.gz emd_17421_validation.pdf.gz | 1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_17421_full_validation.pdf.gz emd_17421_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  emd_17421_validation.xml.gz emd_17421_validation.xml.gz | 16.4 KB | Display | |
| Data in CIF |  emd_17421_validation.cif.gz emd_17421_validation.cif.gz | 21.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17421 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17421 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17421 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17421 | HTTPS FTP |
-Related structure data
| Related structure data |  8p4oMC  8p4mC  8p4nC  8p4pC  8p4rC  8qxsC  8qxtC  8qxuC  8qxvC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17421.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17421.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | GroEL7-GroES7 cage with encapsulated ordered substrate MetK in presence of ADP-BeFx | ||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_17421_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
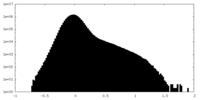
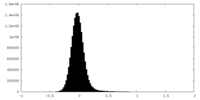
| Density Histograms |
-Half map: #1
| File | emd_17421_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : GroEL14-GroES14-MetK2
| Entire | Name: GroEL14-GroES14-MetK2 |
|---|---|
| Components |
|
-Supramolecule #1: GroEL14-GroES14-MetK2
| Supramolecule | Name: GroEL14-GroES14-MetK2 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 / Details: with bound ADP-BeF3 Mg2+ K+ |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.03 MDa |
-Macromolecule #1: Chaperonin GroEL
| Macromolecule | Name: Chaperonin GroEL / type: protein_or_peptide / ID: 1 / Number of copies: 7 / Enantiomer: LEVO / EC number: chaperonin ATPase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 57.260504 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: AAKDVKFGND ARVKMLRGVN VLADAVKVTL GPKGRNVVLD KSFGAPTITK DGVSVAREIE LEDKFENMGA QMVKEVASKA NDAAGDGTT TATVLAQAII TEGLKAVAAG MNPMDLKRGI DKAVTAAVEE LKALSVPCSD SKAIAQVGTI SANSDETVGK L IAEAMDKV ...String: AAKDVKFGND ARVKMLRGVN VLADAVKVTL GPKGRNVVLD KSFGAPTITK DGVSVAREIE LEDKFENMGA QMVKEVASKA NDAAGDGTT TATVLAQAII TEGLKAVAAG MNPMDLKRGI DKAVTAAVEE LKALSVPCSD SKAIAQVGTI SANSDETVGK L IAEAMDKV GKEGVITVED GTGLQDELDV VEGMQFDRGY LSPYFINKPE TGAVELESPF ILLADKKISN IREMLPVLEA VA KAGKPLL IIAEDVEGEA LATLVVNTMR GIVKVAAVKA PGFGDRRKAM LQDIATLTGG TVISEEIGME LEKATLEDLG QAK RVVINK DTTTIIDGVG EEAAIQGRVA QIRQQIEEAT SDYDREKLQE RVAKLAGGVA VIKVGAATEV EMKEKKARVE DALH ATRAA VEEGVVAGGG VALIRVASKL ADLRGQNEDQ NVGIKVALRA MEAPLRQIVL NCGEEPSVVA NTVKGGDGNY GYNAA TEEY GNMIDMGILD PTKVTRSALQ YAASVAGLMI TTECMVTDLP KNDAADLGAA GGMGGMGGMG GMM UniProtKB: Chaperonin GroEL |
-Macromolecule #2: Co-chaperonin GroES
| Macromolecule | Name: Co-chaperonin GroES / type: protein_or_peptide / ID: 2 / Number of copies: 7 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 10.400938 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MNIRPLHDRV IVKRKEVETK SAGGIVLTGS AAAKSTRGEV LAVGNGRILE NGEVKPLDVK VGDIVIFNDG YGVKSEKIDN EEVLIMSES DILAIVEA UniProtKB: Co-chaperonin GroES |
-Macromolecule #3: S-adenosylmethionine synthase
| Macromolecule | Name: S-adenosylmethionine synthase / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO / EC number: methionine adenosyltransferase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 41.998508 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MAKHLFTSES VSEGHPDKIA DQISDAVLDA ILEQDPKARV ACETYVKTGM VLVGGEITTS AWVDIEEITR NTVREIGYVH SDMGFDANS CAVLSAIGKQ SPDINQGVDR ADPLEQGAGD QGLMFGYATN ETDVLMPAPI TYAHRLVQRQ AEVRKNGTLP W LRPDAKSQ ...String: MAKHLFTSES VSEGHPDKIA DQISDAVLDA ILEQDPKARV ACETYVKTGM VLVGGEITTS AWVDIEEITR NTVREIGYVH SDMGFDANS CAVLSAIGKQ SPDINQGVDR ADPLEQGAGD QGLMFGYATN ETDVLMPAPI TYAHRLVQRQ AEVRKNGTLP W LRPDAKSQ VTFQYDDGKI VGIDAVVLST QHSEEIDQKS LQEAVMEEII KPILPAEWLT SATKFFINPT GRFVIGGPMG DC GLTGRKI IVDTYGGMAR HGGGAFSGKD PSKVDRSAAY AARYVAKNIV AAGLADRCEI QVSYAIGVAE PTSIMVETFG TEK VPSEQL TLLVREFFDL RPYGLIQMLD LLHPIYKETA AYGHFGREHF PWEKTDKAQL LRDAAGLK UniProtKB: S-adenosylmethionine synthase |
-Macromolecule #4: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 4 / Number of copies: 7 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Macromolecule #5: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 5 / Number of copies: 7 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Macromolecule #6: BERYLLIUM TRIFLUORIDE ION
| Macromolecule | Name: BERYLLIUM TRIFLUORIDE ION / type: ligand / ID: 6 / Number of copies: 7 / Formula: BEF |
|---|---|
| Molecular weight | Theoretical: 66.007 Da |
| Chemical component information |  ChemComp-BEF: |
-Macromolecule #7: POTASSIUM ION
| Macromolecule | Name: POTASSIUM ION / type: ligand / ID: 7 / Number of copies: 7 / Formula: K |
|---|---|
| Molecular weight | Theoretical: 39.098 Da |
-Macromolecule #8: water
| Macromolecule | Name: water / type: ligand / ID: 8 / Number of copies: 21 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 10.3 mg/mL | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| ||||||||||||||||||||||||
| Grid | Model: Quantifoil R2/1 / Material: COPPER / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. | ||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: 5.9 mM n-octyl-beta-D-glucopyranoside were added before vitrification. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 55.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 22500 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
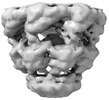
| Initial model |
| ||||||
|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: AB INITIO MODEL | ||||||
| Output model |  PDB-8p4o: |
 Movie
Movie Controller
Controller





























 Z
Z Y
Y X
X