[English] 日本語
 Yorodumi
Yorodumi- EMDB-17407: Structural insights into human co-transcriptional capping - struc... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structural insights into human co-transcriptional capping - structure 5 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | rna polymerase II / capping / TRANSCRIPTION | |||||||||
| Function / homology |  Function and homology information Function and homology informationRNA guanylyltransferase activity / inorganic triphosphate phosphatase activity / negative regulation of DNA-templated transcription, elongation / DSIF complex / regulation of transcription elongation by RNA polymerase II / B-WICH complex positively regulates rRNA expression / RNA Polymerase I Transcription Initiation / RNA Polymerase I Promoter Escape / RNA Polymerase I Transcription Termination / RNA Polymerase III Transcription Initiation From Type 1 Promoter ...RNA guanylyltransferase activity / inorganic triphosphate phosphatase activity / negative regulation of DNA-templated transcription, elongation / DSIF complex / regulation of transcription elongation by RNA polymerase II / B-WICH complex positively regulates rRNA expression / RNA Polymerase I Transcription Initiation / RNA Polymerase I Promoter Escape / RNA Polymerase I Transcription Termination / RNA Polymerase III Transcription Initiation From Type 1 Promoter / RNA Polymerase III Transcription Initiation From Type 2 Promoter / RNA Polymerase III Transcription Initiation From Type 3 Promoter / Formation of RNA Pol II elongation complex / Formation of the Early Elongation Complex / Transcriptional regulation by small RNAs / RNA Polymerase II Pre-transcription Events / TP53 Regulates Transcription of DNA Repair Genes / FGFR2 alternative splicing / RNA polymerase II transcribes snRNA genes / mRNA Capping / mRNA Splicing - Minor Pathway / Processing of Capped Intron-Containing Pre-mRNA / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Elongation / RNA Polymerase II Transcription Initiation And Promoter Clearance / RNA Pol II CTD phosphorylation and interaction with CE / Estrogen-dependent gene expression / Formation of TC-NER Pre-Incision Complex / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / mRNA Splicing - Major Pathway / mRNA 5'-triphosphate monophosphatase activity / mRNA 5'-phosphatase / polynucleotide 5'-phosphatase activity / positive regulation of DNA-templated transcription, elongation / Abortive elongation of HIV-1 transcript in the absence of Tat / transcription elongation-coupled chromatin remodeling / RNA Pol II CTD phosphorylation and interaction with CE during HIV infection / RNA Pol II CTD phosphorylation and interaction with CE / Formation of the Early Elongation Complex / Formation of the HIV-1 Early Elongation Complex / mRNA Capping / maintenance of transcriptional fidelity during transcription elongation by RNA polymerase II / RNA polymerase III activity / Pausing and recovery of Tat-mediated HIV elongation / Tat-mediated HIV elongation arrest and recovery / positive regulation of macroautophagy / phosphoprotein phosphatase activity / positive regulation of nuclear-transcribed mRNA poly(A) tail shortening / HIV elongation arrest and recovery / Pausing and recovery of HIV elongation / RNA polymerase II transcribes snRNA genes / RNA polymerase II activity / Tat-mediated elongation of the HIV-1 transcript / 7-methylguanosine mRNA capping / transcription elongation by RNA polymerase I / Formation of HIV-1 elongation complex containing HIV-1 Tat / transcription-coupled nucleotide-excision repair / tRNA transcription by RNA polymerase III / RNA polymerase I complex / RNA polymerase I activity / RNA polymerase III complex / positive regulation of translational initiation / Formation of HIV elongation complex in the absence of HIV Tat / RNA polymerase II, core complex / RNA Polymerase II Transcription Elongation / RNA processing / Formation of RNA Pol II elongation complex / RNA Polymerase II Pre-transcription Events / translation initiation factor binding / positive regulation of RNA splicing / promoter-specific chromatin binding / TP53 Regulates Transcription of DNA Repair Genes / transcription initiation at RNA polymerase II promoter / transcription elongation by RNA polymerase II / P-body / ribonucleoside binding / fibrillar center / DNA-directed 5'-3' RNA polymerase activity / DNA-directed RNA polymerase / mRNA processing / mRNA guanylyltransferase activity / single-stranded DNA binding / chromosome / Hydrolases; Acting on ester bonds; Exoribonucleases producing 5'-phosphomonoesters / mRNA guanylyltransferase / methyltransferase cap1 / mRNA (nucleoside-2'-O-)-methyltransferase activity / methylation / transcription by RNA polymerase II / nucleic acid binding / chromosome, telomeric region / single-stranded RNA binding / protein dimerization activity / hydrolase activity / nuclear speck / protein heterodimerization activity / RNA-directed RNA polymerase Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Garg G / Dienemann C / Farnung L / Schwarz J / Linden A / Urlaub H / Cramer P | |||||||||
| Funding support | European Union,  Germany, 2 items Germany, 2 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2023 Journal: Mol Cell / Year: 2023Title: Structural insights into human co-transcriptional capping. Authors: Gaurika Garg / Christian Dienemann / Lucas Farnung / Juliane Schwarz / Andreas Linden / Henning Urlaub / Patrick Cramer /  Abstract: Co-transcriptional capping of the nascent pre-mRNA 5' end prevents degradation of RNA polymerase (Pol) II transcripts and suppresses the innate immune response. Here, we provide mechanistic insights ...Co-transcriptional capping of the nascent pre-mRNA 5' end prevents degradation of RNA polymerase (Pol) II transcripts and suppresses the innate immune response. Here, we provide mechanistic insights into the three major steps of human co-transcriptional pre-mRNA capping based on six different cryoelectron microscopy (cryo-EM) structures. The human mRNA capping enzyme, RNGTT, first docks to the Pol II stalk to position its triphosphatase domain near the RNA exit site. The capping enzyme then moves onto the Pol II surface, and its guanylyltransferase receives the pre-mRNA 5'-diphosphate end. Addition of a GMP moiety can occur when the RNA is ∼22 nt long, sufficient to reach the active site of the guanylyltransferase. For subsequent cap(1) methylation, the methyltransferase CMTR1 binds the Pol II stalk and can receive RNA after it is grown to ∼29 nt in length. The observed rearrangements of capping factors on the Pol II surface may be triggered by the completion of catalytic reaction steps and are accommodated by domain movements in the elongation factor DRB sensitivity-inducing factor (DSIF). | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17407.map.gz emd_17407.map.gz | 80.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17407-v30.xml emd-17407-v30.xml emd-17407.xml emd-17407.xml | 35.4 KB 35.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_17407.png emd_17407.png | 97.5 KB | ||
| Filedesc metadata |  emd-17407.cif.gz emd-17407.cif.gz | 11.1 KB | ||
| Others |  emd_17407_half_map_1.map.gz emd_17407_half_map_1.map.gz emd_17407_half_map_2.map.gz emd_17407_half_map_2.map.gz | 80.5 MB 80.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17407 http://ftp.pdbj.org/pub/emdb/structures/EMD-17407 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17407 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17407 | HTTPS FTP |
-Validation report
| Summary document |  emd_17407_validation.pdf.gz emd_17407_validation.pdf.gz | 895.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_17407_full_validation.pdf.gz emd_17407_full_validation.pdf.gz | 895.1 KB | Display | |
| Data in XML |  emd_17407_validation.xml.gz emd_17407_validation.xml.gz | 13.2 KB | Display | |
| Data in CIF |  emd_17407_validation.cif.gz emd_17407_validation.cif.gz | 15.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17407 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17407 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17407 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17407 | HTTPS FTP |
-Related structure data
| Related structure data |  8p4eMC  8p4aC  8p4bC  8p4cC  8p4dC  8p4fC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17407.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17407.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.05 Å | ||||||||||||||||||||||||||||||||||||
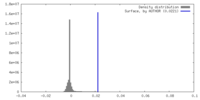
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_17407_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
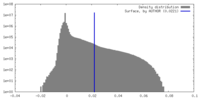
| Density Histograms |
-Half map: #2
| File | emd_17407_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Pol II - TPase complex
+Supramolecule #1: Pol II - TPase complex
+Macromolecule #1: Transcription elongation factor SPT5
+Macromolecule #2: mRNA-capping enzyme
+Macromolecule #3: DNA-directed RNA polymerase II subunit RPB1
+Macromolecule #4: DNA-directed RNA polymerase subunit beta
+Macromolecule #5: DNA-directed RNA polymerase II subunit RPB3
+Macromolecule #6: DNA-directed RNA polymerase II subunit E
+Macromolecule #7: DNA-directed RNA polymerase II subunit F
+Macromolecule #8: DNA-directed RNA polymerases I, II, and III subunit RPABC3
+Macromolecule #9: DNA-directed RNA polymerase II subunit RPB9
+Macromolecule #10: DNA-directed RNA polymerases I, II, and III subunit RPABC5
+Macromolecule #11: DNA-directed RNA polymerase II subunit RPB11-a
+Macromolecule #12: RNA polymerase II subunit K
+Macromolecule #13: RNA polymerase II subunit D
+Macromolecule #14: DNA-directed RNA polymerase II subunit RPB7
+Macromolecule #15: Cap-specific mRNA (nucleoside-2'-O-)-methyltransferase 1
+Macromolecule #16: RNA (5'-D(*(MGT))-R(P*GP*AP*CP*AP*UP*AP*CP*AP*UP*AP*AP*AP*GP*AP*C...
+Macromolecule #17: DNA (26-MER)
+Macromolecule #18: DNA (35-MER)
+Macromolecule #19: MAGNESIUM ION
+Macromolecule #20: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 48148 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller





























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)