+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-13776 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
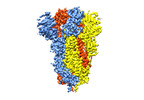
| タイトル | Structure of formaldehyde cross-linked SARS-CoV-2 S glycoprotein | |||||||||
 マップデータ マップデータ | Sharpened 3D map | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | SARS-CoV-2 / glycoprotein / immunization / VIRAL PROTEIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報Maturation of spike protein / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / viral translation / symbiont-mediated-mediated suppression of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / membrane fusion ...Maturation of spike protein / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular space / viral translation / symbiont-mediated-mediated suppression of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / membrane fusion / entry receptor-mediated virion attachment to host cell / Attachment and Entry / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / positive regulation of viral entry into host cell / receptor-mediated virion attachment to host cell / host cell surface receptor binding / symbiont-mediated suppression of host innate immune response / receptor ligand activity / endocytosis involved in viral entry into host cell / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / SARS-CoV-2 activates/modulates innate and adaptive immune responses / host cell plasma membrane / virion membrane / identical protein binding / membrane / plasma membrane 類似検索 - 分子機能 | |||||||||
| 生物種 |  | |||||||||
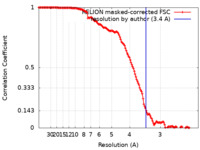
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.4 Å | |||||||||
 データ登録者 データ登録者 | Sulbaran G / Effantin G | |||||||||
| 資金援助 |  フランス, 2件 フランス, 2件
| |||||||||
 引用 引用 |  ジャーナル: Cell Rep Med / 年: 2022 ジャーナル: Cell Rep Med / 年: 2022タイトル: Immunization with synthetic SARS-CoV-2 S glycoprotein virus-like particles protects macaques from infection. 著者: Guidenn Sulbaran / Pauline Maisonnasse / Axelle Amen / Gregory Effantin / Delphine Guilligay / Nathalie Dereuddre-Bosquet / Judith A Burger / Meliawati Poniman / Marloes Grobben / Marlyse ...著者: Guidenn Sulbaran / Pauline Maisonnasse / Axelle Amen / Gregory Effantin / Delphine Guilligay / Nathalie Dereuddre-Bosquet / Judith A Burger / Meliawati Poniman / Marloes Grobben / Marlyse Buisson / Sebastian Dergan Dylon / Thibaut Naninck / Julien Lemaître / Wesley Gros / Anne-Sophie Gallouët / Romain Marlin / Camille Bouillier / Vanessa Contreras / Francis Relouzat / Daphna Fenel / Michel Thepaut / Isabelle Bally / Nicole Thielens / Franck Fieschi / Guy Schoehn / Sylvie van der Werf / Marit J van Gils / Rogier W Sanders / Pascal Poignard / Roger Le Grand / Winfried Weissenhorn /    要旨: The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has caused an ongoing global health crisis. Here, we present as a vaccine candidate synthetic SARS-CoV-2 spike (S) ...The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has caused an ongoing global health crisis. Here, we present as a vaccine candidate synthetic SARS-CoV-2 spike (S) glycoprotein-coated lipid vesicles that resemble virus-like particles. Soluble S glycoprotein trimer stabilization by formaldehyde cross-linking introduces two major inter-protomer cross-links that keep all receptor-binding domains in the "down" conformation. Immunization of cynomolgus macaques with S coated onto lipid vesicles (S-LVs) induces high antibody titers with potent neutralizing activity against the vaccine strain, Alpha, Beta, and Gamma variants as well as T helper (Th)1 CD4-biased T cell responses. Although anti-receptor-binding domain (RBD)-specific antibody responses are initially predominant, the third immunization boosts significant non-RBD antibody titers. Challenging vaccinated animals with SARS-CoV-2 shows a complete protection through sterilizing immunity, which correlates with the presence of nasopharyngeal anti-S immunoglobulin G (IgG) and IgA titers. Thus, the S-LV approach is an efficient and safe vaccine candidate based on a proven classical approach for further development and clinical testing. #2:  ジャーナル: Cell Rep Med / 年: 2022 ジャーナル: Cell Rep Med / 年: 2022タイトル: Immunization with synthetic SARS-CoV-2 S glycoprotein virus-like particles protects macaques from infection. 著者: Sulbaran G / Effantin G / Schoehn G / Weissenhorn W | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_13776.map.gz emd_13776.map.gz | 216.6 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-13776-v30.xml emd-13776-v30.xml emd-13776.xml emd-13776.xml | 18.7 KB 18.7 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_13776_fsc.xml emd_13776_fsc.xml | 14.2 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_13776.png emd_13776.png | 73 KB | ||
| Filedesc metadata |  emd-13776.cif.gz emd-13776.cif.gz | 7 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13776 http://ftp.pdbj.org/pub/emdb/structures/EMD-13776 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13776 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13776 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_13776_validation.pdf.gz emd_13776_validation.pdf.gz | 366.2 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_13776_full_validation.pdf.gz emd_13776_full_validation.pdf.gz | 365.8 KB | 表示 | |
| XML形式データ |  emd_13776_validation.xml.gz emd_13776_validation.xml.gz | 13.8 KB | 表示 | |
| CIF形式データ |  emd_13776_validation.cif.gz emd_13776_validation.cif.gz | 18.9 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13776 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13776 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13776 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-13776 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_13776.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_13776.map.gz / 形式: CCP4 / 大きさ: 244.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Sharpened 3D map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.15 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : Trimer of the SARS_CoV-2 S glycoprotein
| 全体 | 名称: Trimer of the SARS_CoV-2 S glycoprotein |
|---|---|
| 要素 |
|
-超分子 #1: Trimer of the SARS_CoV-2 S glycoprotein
| 超分子 | 名称: Trimer of the SARS_CoV-2 S glycoprotein / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1 |
|---|---|
| 由来(天然) | 生物種:  |
-分子 #1: Spike glycoprotein
| 分子 | 名称: Spike glycoprotein / タイプ: protein_or_peptide / ID: 1 / コピー数: 3 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 142.399375 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS NVTWFHAIHV SGTNGTKRFD NPVLPFNDG VYFASTEKSN IIRGWIFGTT LDSKTQSLLI VNNATNVVIK VCEFQFCNDP FLGVYYHKNN KSWMESEFRV Y SSANNCTF ...文字列: MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS NVTWFHAIHV SGTNGTKRFD NPVLPFNDG VYFASTEKSN IIRGWIFGTT LDSKTQSLLI VNNATNVVIK VCEFQFCNDP FLGVYYHKNN KSWMESEFRV Y SSANNCTF EYVSQPFLMD LEGKQGNFKN LREFVFKNID GYFKIYSKHT PINLVRDLPQ GFSALEPLVD LPIGINITRF QT LLALHRS YLTPGDSSSG WTAGAAAYYV GYLQPRTFLL KYNENGTITD AVDCALDPLS ETKCTLKSFT VEKGIYQTSN FRV QPTESI VRFPNITNLC PFGEVFNATR FASVYAWNRK RISNCVADYS VLYNSASFST FKCYGVSPTK LNDLCFTNVY ADSF VIRGD EVRQIAPGQT GKIADYNYKL PDDFTGCVIA WNSNNLDSKV GGNYNYLYRL FRKSNLKPFE RDISTEIYQA GSTPC NGVE GFNCYFPLQS YGFQPTNGVG YQPYRVVVLS FELLHAPATV CGPKKSTNLV KNKCVNFNFN GLTGTGVLTE SNKKFL PFQ QFGRDIADTT DAVRDPQTLE ILDITPCSFG GVSVITPGTN TSNQVAVLYQ DVNCTEVPVA IHADQLTPTW RVYSTGS NV FQTRAGCLIG AEHVNNSYEC DIPIGAGICA SYQTQTNSPG SASSVASQSI IAYTMSLGAE NSVAYSNNSI AIPTNFTI S VTTEILPVSM TKTSVDCTMY ICGDSTECSN LLLQYGSFCT QLNRALTGIA VEQDKNTQEV FAQVKQIYKT PPIKDFGGF NFSQILPDPS KPSKRSFIED LLFNKVTLAD AGFIKQYGDC LGDIAARDLI CAQKFNGLTV LPPLLTDEMI AQYTSALLAG TITSGWTFG AGAALQIPFA MQMAYRFNGI GVTQNVLYEN QKLIANQFNS AIGKIQDSLS STASALGKLQ DVVNQNAQAL N TLVKQLSS NFGAISSVLN DILSRLDPPE AEVQIDRLIT GRLQSLQTYV TQQLIRAAEI RASANLAATK MSECVLGQSK RV DFCGKGY HLMSFPQSAP HGVVFLHVTY VPAQEKNFTT APAICHDGKA HFPREGVFVS NGTHWFVTQR NFYEPQIITT DNT FVSGNC DVVIGIVNNT VYDPLQPELD SFKEELDKYF KNHTSPDVDL GDISGINASV VNIQKEIDRL NEVAKNLNES LIDL QELGK YEQGSGYIPE APRDGQAYVR KDGEWVLLST FLGRSLEVLF QGPGHHHHHH HHSAWSHPQF EKGGGSGGGG SGGSA WSHP QFEK UniProtKB: Spike glycoprotein |
-分子 #3: 2-acetamido-2-deoxy-beta-D-glucopyranose
| 分子 | 名称: 2-acetamido-2-deoxy-beta-D-glucopyranose / タイプ: ligand / ID: 3 / コピー数: 33 / 式: NAG |
|---|---|
| 分子量 | 理論値: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | TFS GLACIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: COUNTING / 平均電子線量: 40.0 e/Å2 |
| 電子線 | 加速電圧: 200 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
 ムービー
ムービー コントローラー
コントローラー


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)