+Search query
-Structure paper
| Title | Structures of human SGLT in the occluded state reveal conformational changes during sugar transport. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 14, Issue 1, Page 2920, Year 2023 |
| Publish date | May 22, 2023 |
 Authors Authors | Wenhao Cui / Yange Niu / Zejian Sun / Rui Liu / Lei Chen /  |
| PubMed Abstract | Sodium-Glucose Cotransporters (SGLT) mediate the uphill uptake of extracellular sugars and play fundamental roles in sugar metabolism. Although their structures in inward-open and outward-open ...Sodium-Glucose Cotransporters (SGLT) mediate the uphill uptake of extracellular sugars and play fundamental roles in sugar metabolism. Although their structures in inward-open and outward-open conformations are emerging from structural studies, the trajectory of how SGLTs transit from the outward-facing to the inward-facing conformation remains unknown. Here, we present the cryo-EM structures of human SGLT1 and SGLT2 in the substrate-bound state. Both structures show an occluded conformation, with not only the extracellular gate but also the intracellular gate tightly sealed. The sugar substrate are caged inside a cavity surrounded by TM1, TM2, TM3, TM6, TM7, and TM10. Further structural analysis reveals the conformational changes associated with the binding and release of substrates. These structures fill a gap in our understanding of the structural mechanisms of SGLT transporters. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:37217492 / PubMed:37217492 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.26 - 3.48 Å |
| Structure data | EMDB-33962, PDB-7yni: EMDB-33963, PDB-7ynj: EMDB-33964, PDB-7ynk: |
| Chemicals |  ChemComp-KQC:  ChemComp-GYP: |
| Source |
|
 Keywords Keywords | PROTEIN TRANSPORT / glucose transporter / SGLT / sodium glucose transporter / membrane protein |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



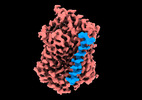





 homo sapiens (human)
homo sapiens (human)