+Search query
-Structure paper
| Title | High-resolution snapshots of human N-myristoyltransferase in action illuminate a mechanism promoting N-terminal Lys and Gly myristoylation. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 11, Page 1132-1132, Year 2020 |
| Publish date | Sep 13, 2017 (structure data deposition date) |
 Authors Authors | Dian, C. / Perez-Dorado, I. / Riviere, F. / Asensio, T. / Legrand, P. / Ritzefeld, M. / Shen, M. / Cota, E. / Meinnel, T. / Tate, E.W. / Giglione, C. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:32111831 PubMed:32111831 |
| Methods | X-ray diffraction |
| Resolution | 1.87 - 2.8 Å |
| Structure data |  PDB-6ehj:  PDB-6qrm:  PDB-6sjz:  PDB-6sk2:  PDB-6sk3:  PDB-6sk8:  PDB-6skj: |
| Chemicals |  ChemComp-GOL:  ChemComp-MYA:  ChemComp-GLY:  ChemComp-SER: 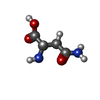 ChemComp-ASN:  ChemComp-LYS: 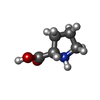 ChemComp-PRO:  ChemComp-MYR:  ChemComp-COA:  ChemComp-HOH:  ChemComp-CL:  ChemComp-MG: |
| Source |
|
 Keywords Keywords | TRANSFERASE / Myristoylation / complex / substrate peptide / Myristoyltransferase type1 / Acyltransferase / GNAT / GCN5-Related N-Acetyltransferases / NMT / E-MYRISTOYLATION |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers



 homo sapiens (human)
homo sapiens (human)