+Search query
-Structure paper
| Title | Structural insight into substrate and inhibitor discrimination by human P-glycoprotein. |
|---|---|
| Journal, issue, pages | Science, Vol. 363, Issue 6428, Page 753-756, Year 2019 |
| Publish date | Feb 15, 2019 |
 Authors Authors | Amer Alam / Julia Kowal / Eugenia Broude / Igor Roninson / Kaspar P Locher /   |
| PubMed Abstract | ABCB1, also known as P-glycoprotein, actively extrudes xenobiotic compounds across the plasma membrane of diverse cells, which contributes to cellular drug resistance and interferes with therapeutic ...ABCB1, also known as P-glycoprotein, actively extrudes xenobiotic compounds across the plasma membrane of diverse cells, which contributes to cellular drug resistance and interferes with therapeutic drug delivery. We determined the 3.5-angstrom cryo-electron microscopy structure of substrate-bound human ABCB1 reconstituted in lipidic nanodiscs, revealing a single molecule of the chemotherapeutic compound paclitaxel (Taxol) bound in a central, occluded pocket. A second structure of inhibited, human-mouse chimeric ABCB1 revealed two molecules of zosuquidar occupying the same drug-binding pocket. Minor structural differences between substrate- and inhibitor-bound ABCB1 sites are amplified toward the nucleotide-binding domains (NBDs), revealing how the plasticity of the drug-binding site controls the dynamics of the adenosine triphosphate-hydrolyzing NBDs. Ordered cholesterol and phospholipid molecules suggest how the membrane modulates the conformational changes associated with drug binding and transport. |
 External links External links |  Science / Science /  PubMed:30765569 / PubMed:30765569 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.5 - 3.9 Å |
| Structure data | EMDB-4536, PDB-6qee: EMDB-4539, PDB-6qex:  EMDB-4540:  EMDB-4541: |
| Chemicals |  ChemComp-NAG: 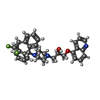 ChemComp-ZQU:  ChemComp-CLR:  ChemComp-3PE:  ChemComp-TA1: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / ABCB1 / p-glycoprotein / p-gp / multidrug transporter / ABC transporter / zosuquidar / membrane transporter / multidrug exporter |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 homo sapiens (human)
homo sapiens (human)
