+Search query
-Structure paper
| Title | Post-catalysis structures of mitochondrial complex I with ubiquinol-10 bound in the active site. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 17, Issue 1, Year 2026 |
| Publish date | Mar 5, 2026 |
 Authors Authors | Injae Chung / Caroline S Pereira / John J Wright / Guilherme M Arantes / Judy Hirst /    |
| PubMed Abstract | Respiratory complex I is a multi-subunit energy-transducing membrane enzyme essential for mitochondrial and cellular energy metabolism. It couples NADH oxidation and ubiquinone-10 (Q) reduction to ...Respiratory complex I is a multi-subunit energy-transducing membrane enzyme essential for mitochondrial and cellular energy metabolism. It couples NADH oxidation and ubiquinone-10 (Q) reduction to the concomitant pumping of four protons to generate the proton-motive force that powers oxidative phosphorylation. Despite recent advances in structural knowledge of complex I, many mechanistic aspects including the reactive binding poses of Q, how Q reduction initiates the proton transfer cascade, and how protons move through the membrane domain, remain unclear. Here, we use electron cryomicroscopy to determine structures of mammalian complex I, reconstituted into phospholipid nanodiscs containing exogenous Q and reduced by NADH, to global resolutions of 2.0 to 2.6 Å. Two conformations of a reduced QH molecule are observed, fully inserted into the Q-binding channel in the turnover-relevant closed state. By comparing the quinone species bound in oxidised and reduced complex I structures, paired with molecular dynamics simulations to investigate the charge states of key surrounding residues, we propose a series of substrate binding poses that Q transits through for reduction. Our highly hydrated structures exhibit near-continuous proton-transfer connections along the length of the membrane domain, enabling comparisons between them to assist in identifying the proton-transfer control points that are essential to catalysis. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:41786699 / PubMed:41786699 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.01 - 2.61 Å |
| Structure data | EMDB-55030, PDB-9smf: EMDB-55031, PDB-9smg: EMDB-55032, PDB-9smh: EMDB-55033, PDB-9smi:  EMDB-55034: Reduced bovine complex I in lipid nanodisc, NADH-slack |
| Chemicals |  ChemComp-3PE:  ChemComp-PC1:  ChemComp-SF4:  ChemComp-U10:  ChemComp-FES:  ChemComp-FMN:  ChemComp-NAI:  ChemComp-K:  ChemComp-CDL: 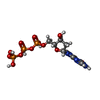 ChemComp-DGT:  ChemComp-MG:  ChemComp-NDP:  ChemComp-ZN:  ChemComp-EHZ: 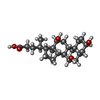 ChemComp-CHD:  ChemComp-MYR:  ChemComp-HOH:  ChemComp-LMT: |
| Source |
|
 Keywords Keywords | ELECTRON TRANSPORT / Mitochondrial complex I / Respiratory complex I / NADH:ubiquinone oxidoreductase / Ubiquinone / Nanodisc / NADH |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers












