+Search query
-Structure paper
| Title | The CryoEM structure of human serum albumin in complex with ligands. |
|---|---|
| Journal, issue, pages | J Struct Biol, Vol. 216, Issue 3, Page 108105, Year 2024 |
| Publish date | Jun 8, 2024 |
 Authors Authors | Claudio Catalano / Kyle W Lucier / Dennis To / Skerdi Senko / Nhi L Tran / Ashlyn C Farwell / Sabrina M Silva / Phat V Dip / Nicole Poweleit / Giovanna Scapin /  |
| PubMed Abstract | Human serum albumin (HSA) is the most prevalent plasma protein in the human body, accounting for 60 % of the total plasma protein. HSA plays a major pharmacokinetic function, serving as a ...Human serum albumin (HSA) is the most prevalent plasma protein in the human body, accounting for 60 % of the total plasma protein. HSA plays a major pharmacokinetic function, serving as a facilitator in the distribution of endobiotics and xenobiotics within the organism. In this paper we report the cryoEM structures of HSA in the apo form and in complex with two ligands (salicylic acid and teniposide) at a resolution of 3.5, 3.7 and 3.4 Å, respectively. We expand upon previously published work and further demonstrate that sub-4 Å maps of ∼60 kDa proteins can be routinely obtained using a 200 kV microscope, employing standard workflows. Most importantly, these maps allowed for the identification of small molecule ligands, emphasizing the practical applicability of this methodology and providing a starting point for subsequent computational modeling and in silico optimization. |
 External links External links |  J Struct Biol / J Struct Biol /  PubMed:38852682 PubMed:38852682 |
| Methods | EM (single particle) |
| Resolution | 3.4 - 3.7 Å |
| Structure data | EMDB-43088, PDB-8vac: EMDB-43089, PDB-8vae: EMDB-43090, PDB-8vaf: |
| Chemicals |  ChemComp-9TP: 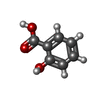 ChemComp-SAL: |
| Source |
|
 Keywords Keywords | TRANSPORT PROTEIN / Human Serum Albumin / teniposide / Salicylic acid |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers









 homo sapiens (human)
homo sapiens (human)