+Search query
-Structure paper
| Title | Stepwise activation of a metabotropic glutamate receptor. |
|---|---|
| Journal, issue, pages | Nature, Vol. 629, Issue 8013, Page 951-956, Year 2024 |
| Publish date | Apr 17, 2024 |
 Authors Authors | Kaavya Krishna Kumar / Haoqing Wang / Chris Habrian / Naomi R Latorraca / Jun Xu / Evan S O'Brien / Chensong Zhang / Elizabeth Montabana / Antoine Koehl / Susan Marqusee / Ehud Y Isacoff / Brian K Kobilka /  |
| PubMed Abstract | Metabotropic glutamate receptors belong to a family of G protein-coupled receptors that are obligate dimers and possess a large extracellular ligand-binding domain that is linked via a cysteine-rich ...Metabotropic glutamate receptors belong to a family of G protein-coupled receptors that are obligate dimers and possess a large extracellular ligand-binding domain that is linked via a cysteine-rich domain to their 7-transmembrane domain. Upon activation, these receptors undergo a large conformational change to transmit the ligand binding signal from the extracellular ligand-binding domain to the G protein-coupling 7-transmembrane domain. In this manuscript, we propose a model for a sequential, multistep activation mechanism of metabotropic glutamate receptor subtype 5. We present a series of structures in lipid nanodiscs, from inactive to fully active, including agonist-bound intermediate states. Further, using bulk and single-molecule fluorescence imaging, we reveal distinct receptor conformations upon allosteric modulator and G protein binding. |
 External links External links |  Nature / Nature /  PubMed:38632403 / PubMed:38632403 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.89 - 4.65 Å |
| Structure data | EMDB-41069, PDB-8t6j: EMDB-41092, PDB-8t7h: EMDB-41099, PDB-8t8m: EMDB-41139, PDB-8tao:  EMDB-41783: CDPPB bound inactive mGlu5, local refinement on TM region  EMDB-41787: CDPPB bound inactive mGlu5, non-uniform refinement of the whole complex  EMDB-41789: TM region local refinement of CDPPB, QUS bound mGlu5 in complex with Nb43  EMDB-41793: CDDPPB, QUS bound mGlu5 in complex with Nb43, local refinement on the VFT region  EMDB-41804: QUS bound active mGlu5 in complex with Nb43, local refinement on the TM region  EMDB-41807: QUS bounds mGlu5 in complex with Nb43, non uniform refinement on the whole complex  EMDB-41808: QUS bound inactive mGlu5 in complex with Nb43, local refinement on the TM region |
| Chemicals | 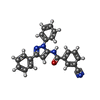 ChemComp-YKU: 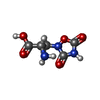 ChemComp-QUS: |
| Source |
|
 Keywords Keywords | SIGNALING PROTEIN / GPCR |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers











 homo sapiens (human)
homo sapiens (human)
