+Search query
-Structure paper
| Title | DPP9 sequesters the C terminus of NLRP1 to repress inflammasome activation. |
|---|---|
| Journal, issue, pages | Nature, Vol. 592, Issue 7856, Page 778-783, Year 2021 |
| Publish date | Mar 17, 2021 |
 Authors Authors | L Robert Hollingsworth / Humayun Sharif / Andrew R Griswold / Pietro Fontana / Julian Mintseris / Kevin B Dagbay / Joao A Paulo / Steven P Gygi / Daniel A Bachovchin / Hao Wu /  |
| PubMed Abstract | Nucleotide-binding domain and leucine-rich repeat pyrin-domain containing protein 1 (NLRP1) is an inflammasome sensor that mediates the activation of caspase-1 to induce cytokine maturation and ...Nucleotide-binding domain and leucine-rich repeat pyrin-domain containing protein 1 (NLRP1) is an inflammasome sensor that mediates the activation of caspase-1 to induce cytokine maturation and pyroptosis. Gain-of-function mutations of NLRP1 cause severe inflammatory diseases of the skin. NLRP1 contains a function-to-find domain that auto-proteolyses into noncovalently associated subdomains, and proteasomal degradation of the repressive N-terminal fragment of NLRP1 releases its inflammatory C-terminal fragment (NLRP1 CT). Cytosolic dipeptidyl peptidases 8 and 9 (hereafter, DPP8/DPP9) both interact with NLRP1, and small-molecule inhibitors of DPP8/DPP9 activate NLRP1 by mechanisms that are currently unclear. Here we report cryo-electron microscopy structures of the human NLRP1-DPP9 complex alone and with Val-boroPro (VbP), an inhibitor of DPP8/DPP9. The structures reveal a ternary complex that comprises DPP9, full-length NLRP1 and the NLRPT CT. The binding of the NLRP1 CT to DPP9 requires full-length NLRP1, which suggests that NLRP1 activation is regulated by the ratio of NLRP1 CT to full-length NLRP1. Activation of the inflammasome by ectopic expression of the NLRP1 CT is consistently rescued by co-expression of autoproteolysis-deficient full-length NLRP1. The N terminus of the NLRP1 CT inserts into the DPP9 active site, and VbP disrupts this interaction. Thus, VbP weakens the NLRP1-DPP9 interaction and accelerates degradation of the N-terminal fragment to induce inflammasome activation. Overall, these data demonstrate that DPP9 quenches low levels of NLRP1 CT and thus serves as a checkpoint for activation of the NLRP1 inflammasome. |
 External links External links |  Nature / Nature /  PubMed:33731932 / PubMed:33731932 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 2.9 - 3.6 Å |
| Structure data | EMDB-22074, PDB-6x6a: EMDB-22075, PDB-6x6c: |
| Chemicals | 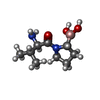 ChemComp-GK2: |
| Source |
|
 Keywords Keywords | IMMUNE SYSTEM / NLRP1 / DPP9 / inflammasome / Val-boroPro (VbP) / talabostat / innate immunity |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 homo sapiens (human)
homo sapiens (human)