+検索条件
-Structure paper
| タイトル | Structure-guided mutagenesis of OSCAs reveals differential activation to mechanical stimuli. |
|---|---|
| ジャーナル・号・ページ | Elife, Vol. 12, Year 2024 |
| 掲載日 | 2024年4月9日 |
 著者 著者 | Sebastian Jojoa-Cruz / Adrienne E Dubin / Wen-Hsin Lee / Andrew B Ward /  |
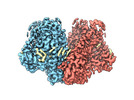
| PubMed 要旨 | The dimeric two-pore OSCA/TMEM63 family has recently been identified as mechanically activated ion channels. Previously, based on the unique features of the structure of OSCA1.2, we postulated the ...The dimeric two-pore OSCA/TMEM63 family has recently been identified as mechanically activated ion channels. Previously, based on the unique features of the structure of OSCA1.2, we postulated the potential involvement of several structural elements in sensing membrane tension (Jojoa-Cruz et al., 2018). Interestingly, while OSCA1, 2, and 3 clades are activated by membrane stretch in cell-attached patches (i.e. they are stretch-activated channels), they differ in their ability to transduce membrane deformation induced by a blunt probe (poking). Here, in an effort to understand the domains contributing to mechanical signal transduction, we used cryo-electron microscopy to solve the structure of (At) OSCA3.1, which, unlike AtOSCA1.2, only produced stretch- but not poke-activated currents in our initial characterization (Murthy et al., 2018). Mutagenesis and electrophysiological assessment of conserved and divergent putative mechanosensitive features of OSCA1.2 reveal a selective disruption of the macroscopic currents elicited by poking without considerable effects on stretch-activated currents (SAC). Our results support the involvement of the amphipathic helix and lipid-interacting residues in the membrane fenestration in the response to poking. Our findings position these two structural elements as potential sources of functional diversity within the family. |
 リンク リンク |  Elife / Elife /  PubMed:38592763 / PubMed:38592763 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.6 Å |
| 構造データ | EMDB-41911, PDB-8u53: |
| 化合物 |  ChemComp-CPL:  ChemComp-82T:  ChemComp-PLM: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Mechanically activated ion channel |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について