+検索条件
-Structure paper
| タイトル | A selectivity filter in the ER membrane protein complex limits protein misinsertion at the ER. |
|---|---|
| ジャーナル・号・ページ | J Cell Biol, Vol. 222, Issue 8, Year 2023 |
| 掲載日 | 2023年8月7日 |
 著者 著者 | Tino Pleiner / Masami Hazu / Giovani Pinton Tomaleri / Vy N Nguyen / Kurt Januszyk / Rebecca M Voorhees /  |
| PubMed 要旨 | Tail-anchored (TA) proteins play essential roles in mammalian cells, and their accurate localization is critical for proteostasis. Biophysical similarities lead to mistargeting of mitochondrial TA ...Tail-anchored (TA) proteins play essential roles in mammalian cells, and their accurate localization is critical for proteostasis. Biophysical similarities lead to mistargeting of mitochondrial TA proteins to the ER, where they are delivered to the insertase, the ER membrane protein complex (EMC). Leveraging an improved structural model of the human EMC, we used mutagenesis and site-specific crosslinking to map the path of a TA protein from its cytosolic capture by methionine-rich loops to its membrane insertion through a hydrophilic vestibule. Positively charged residues at the entrance to the vestibule function as a selectivity filter that uses charge-repulsion to reject mitochondrial TA proteins. Similarly, this selectivity filter retains the positively charged soluble domains of multipass substrates in the cytosol, thereby ensuring they adopt the correct topology and enforcing the "positive-inside" rule. Substrate discrimination by the EMC provides a biochemical explanation for one role of charge in TA protein sorting and protects compartment integrity by limiting protein misinsertion. |
 リンク リンク |  J Cell Biol / J Cell Biol /  PubMed:37199759 / PubMed:37199759 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.5 - 3.9 Å |
| 構造データ | EMDB-40245: Human ER membrane protein complex (EMC) in GDN, 9-subunit map 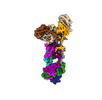 EMDB-40246: Human ER membrane protein complex (EMC) in GDN, Consensus map 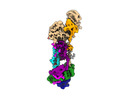 EMDB-40247: Human ER membrane protein complex (EMC) in GDN, 8-subunit map |
| 化合物 |  ChemComp-NAG:  ChemComp-PCW: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Insertase / endoplasmic reticulum / transmembrane chaperone |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト)