+検索条件
-Structure paper
| タイトル | A robust approach for MicroED sample preparation of lipidic cubic phase embedded membrane protein crystals. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 14, Issue 1, Page 1086, Year 2023 |
| 掲載日 | 2023年2月25日 |
 著者 著者 | Michael W Martynowycz / Anna Shiriaeva / Max T B Clabbers / William J Nicolas / Sara J Weaver / Johan Hattne / Tamir Gonen /  |
| PubMed 要旨 | Crystallizing G protein-coupled receptors (GPCRs) in lipidic cubic phase (LCP) often yields crystals suited for the cryogenic electron microscopy (cryoEM) method microcrystal electron diffraction ...Crystallizing G protein-coupled receptors (GPCRs) in lipidic cubic phase (LCP) often yields crystals suited for the cryogenic electron microscopy (cryoEM) method microcrystal electron diffraction (MicroED). However, sample preparation is challenging. Embedded crystals cannot be targeted topologically. Here, we use an integrated fluorescence light microscope (iFLM) inside of a focused ion beam and scanning electron microscope (FIB-SEM) to identify fluorescently labeled GPCR crystals. Crystals are targeted using the iFLM and LCP is milled using a plasma focused ion beam (pFIB). The optimal ion source for preparing biological lamellae is identified using standard crystals of proteinase K. Lamellae prepared using either argon or xenon produced the highest quality data and structures. MicroED data are collected from the milled lamellae and the structures are determined. This study outlines a robust approach to identify and mill membrane protein crystals for MicroED and demonstrates plasma ion-beam milling is a powerful tool for preparing biological lamellae. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:36841804 / PubMed:36841804 /  PubMed Central PubMed Central |
| 手法 | EM (電子線結晶学) |
| 解像度 | 1.39 - 2.0 Å |
| 構造データ | EMDB-29586, PDB-8fyn: EMDB-29587, PDB-8fyo: EMDB-29588, PDB-8fyp: EMDB-29590, PDB-8fyq: EMDB-29595, PDB-8fyr: EMDB-29596, PDB-8fys: |
| 化合物 | 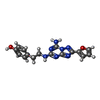 ChemComp-ZMA:  ChemComp-CLR:  ChemComp-OLA:  ChemComp-OLC: 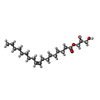 ChemComp-OLB:  ChemComp-NA:  ChemComp-HOH:  ChemComp-NO3:  ChemComp-CA: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / A2A Adenosine Receptor / HYDROLASE |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について















 homo sapiens (ヒト)
homo sapiens (ヒト) parengyodontium album (菌類)
parengyodontium album (菌類)