+検索条件
-Structure paper
| タイトル | CryoEM Structure with ATP Synthase Enables Late-Stage Diversification of Cruentaren A. |
|---|---|
| ジャーナル・号・ページ | Chemistry, Vol. 29, Issue 29, Page e202300262, Year 2023 |
| 掲載日 | 2023年5月22日 |
 著者 著者 | Xiaozheng Dou / Hui Guo / Terin D'Amico / Leah Abdallah / Chitra Subramanian / Bhargav A Patel / Mark Cohen / John L Rubinstein / Brian S J Blagg /   |
| PubMed 要旨 | Cruentaren A is a natural product that exhibits potent antiproliferative activity against various cancer cell lines, yet its binding site within ATP synthase remained unknown, thus limiting the ...Cruentaren A is a natural product that exhibits potent antiproliferative activity against various cancer cell lines, yet its binding site within ATP synthase remained unknown, thus limiting the development of improved analogues as anticancer agents. Herein, we report the cryogenic electron microscopy (cryoEM) structure of cruentaren A bound to ATP synthase, which allowed the design of new inhibitors through semisynthetic modification. Examples of cruentaren A derivatives include a trans-alkene isomer, which was found to exhibit similar activity to cruentaren A against three cancer cell lines as well as several other analogues that retained potent inhibitory activity. Together, these studies provide a foundation for the generation of cruentaren A derivatives as potential therapeutics for the treatment of cancer. |
 リンク リンク |  Chemistry / Chemistry /  PubMed:36867738 / PubMed:36867738 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.9 - 4.5 Å |
| 構造データ | EMDB-28818, PDB-8f2k:  EMDB-28819: Structure of yeast F1-ATPase determined with 25 micromolar cruentaren A |
| 化合物 |  ChemComp-ATP:  ChemComp-MG: 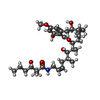 ChemComp-XBC: |
| 由来 |
|
 キーワード キーワード | HYDROLASE / F1-ATPase / ATP Synthase / cruentaren A / drug development |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について






