+Search query
-Structure paper
| Title | An allosteric transport mechanism for the AcrAB-TolC multidrug efflux pump. |
|---|---|
| Journal, issue, pages | Elife, Vol. 6, Year 2017 |
| Publish date | Mar 29, 2017 |
 Authors Authors | Zhao Wang / Guizhen Fan / Corey F Hryc / James N Blaza / Irina I Serysheva / Michael F Schmid / Wah Chiu / Ben F Luisi / Dijun Du /   |
| PubMed Abstract | Bacterial efflux pumps confer multidrug resistance by transporting diverse antibiotics from the cell. In Gram-negative bacteria, some of these pumps form multi-protein assemblies that span the cell ...Bacterial efflux pumps confer multidrug resistance by transporting diverse antibiotics from the cell. In Gram-negative bacteria, some of these pumps form multi-protein assemblies that span the cell envelope. Here, we report the near-atomic resolution cryoEM structures of the AcrAB-TolC multidrug efflux pump in resting and drug transport states, revealing a quaternary structural switch that allosterically couples and synchronizes initial ligand binding with channel opening. Within the transport-activated state, the channel remains open even though the pump cycles through three distinct conformations. Collectively, our data provide a dynamic mechanism for the assembly and operation of the AcrAB-TolC pump. |
 External links External links |  Elife / Elife /  PubMed:28355133 / PubMed:28355133 /  PubMed Central PubMed Central |
| Methods | EM (single particle) / X-ray diffraction |
| Resolution | 3.2 - 6.5 Å |
| Structure data | EMDB-3636, PDB-5ng5: EMDB-8636, PDB-5v5s: EMDB-8640: Asymmetric structure of AcrAB-TolC tripartite multi drug efflux pump  PDB-5nc5: |
| Chemicals |  ChemComp-LMT:  ChemComp-D12:  ChemComp-D10: 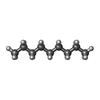 ChemComp-DD9: 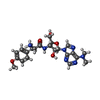 ChemComp-PUY: 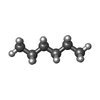 ChemComp-HEX:  ChemComp-HOH: 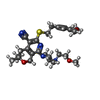 ChemComp-5QF: |
| Source |
|
 Keywords Keywords | TRANSPORT PROTEIN / Membrane transporter / Multidrug efflux pump / MEMBRANE PROTEIN / multi-drug efflux; membrane transport; RND superfamily; Drug resistance / multi-drug efflux / membrane transport / RND superfamily / Drug resistance |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers










