+Search query
-Structure paper
| Title | Activation of the α adrenoceptor by the sedative sympatholytic dexmedetomidine. |
|---|---|
| Journal, issue, pages | Nat Chem Biol, Vol. 16, Issue 5, Page 507-512, Year 2020 |
| Publish date | Mar 9, 2020 |
 Authors Authors | Daopeng Yuan / Zhongmin Liu / Jonas Kaindl / Shoji Maeda / Jiawei Zhao / Xiaoou Sun / Jun Xu / Peter Gmeiner / Hong-Wei Wang / Brian K Kobilka /    |
| PubMed Abstract | The α adrenergic receptors (αARs) are G protein-coupled receptors (GPCRs) that respond to adrenaline and noradrenaline and couple to the Gi/o family of G proteins. αARs play important roles in ...The α adrenergic receptors (αARs) are G protein-coupled receptors (GPCRs) that respond to adrenaline and noradrenaline and couple to the Gi/o family of G proteins. αARs play important roles in regulating the sympathetic nervous system. Dexmedetomidine is a highly selective αAR agonist used in post-operative patients as an anxiety-reducing, sedative medicine that decreases the requirement for opioids. As is typical for selective αAR agonists, dexmedetomidine consists of an imidazole ring and a substituted benzene moiety lacking polar groups, which is in contrast to βAR-selective agonists, which share an ethanolamine group and an aromatic system with polar, hydrogen-bonding substituents. To better understand the structural basis for the selectivity and efficacy of adrenergic agonists, we determined the structure of the αAR in complex with dexmedetomidine and Go at a resolution of 2.9 Å by single-particle cryo-EM. The structure reveals the mechanism of αAR-selective activation and provides insights into Gi/o coupling specificity. |
 External links External links |  Nat Chem Biol / Nat Chem Biol /  PubMed:32152538 PubMed:32152538 |
| Methods | EM (single particle) |
| Resolution | 2.9 - 4.1 Å |
| Structure data | |
| Chemicals | 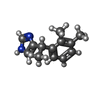 ChemComp-CZX: |
| Source |
|
 Keywords Keywords | MEMBRANE PROTEIN / GPCR / Complex / cryo-EM |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 Spodoptera (butterflies/moths)
Spodoptera (butterflies/moths) homo sapiens (human)
homo sapiens (human)
 enterobacteria phage rb59 (virus)
enterobacteria phage rb59 (virus)