+Search query
-Structure paper
| Title | Cryo-EM structure of the Agrobacterium tumefaciens T-pilus reveals the importance of positive charges in the lumen. |
|---|---|
| Journal, issue, pages | Structure, Vol. 31, Issue 4, Page 375-384.e4, Year 2023 |
| Publish date | Apr 6, 2023 |
 Authors Authors | Jaafar Amro / Corbin Black / Zakaria Jemouai / Nathan Rooney / Caroline Daneault / Natalie Zeytuni / Matthieu Ruiz / Khanh Huy Bui / Christian Baron /  |
| PubMed Abstract | Agrobacterium tumefaciens is a natural genetic engineer that transfers DNA into plants, which is the most applied process for generation of genetically modified plants. DNA transfer is mediated by a ...Agrobacterium tumefaciens is a natural genetic engineer that transfers DNA into plants, which is the most applied process for generation of genetically modified plants. DNA transfer is mediated by a type IV secretion system in the cell envelope and extracellular T-pili. We here report the cryo-electron microscopic structures of the T-pilus at 3.2-Å resolution and of the plasmid pKM101-determined N-pilus at 3-Å resolution. Both pili contain a main pilus protein (VirB2 in A. tumefaciens, TraM in pKM101) and phospholipids arranged in a five-start helical assembly. They contain positively charged amino acids in the lumen, and the lipids are positively charged in the T-pilus (phosphatidylcholine) conferring overall positive charge. Mutagenesis of the lumen-exposed Arg91 in VirB2 results in protein destabilization and loss of pilus formation. Our results reveal that different phospholipids can be incorporated into type IV secretion pili and that the charge of the lumen may be of functional importance. |
 External links External links |  Structure / Structure /  PubMed:36513067 PubMed:36513067 |
| Methods | EM (single particle) |
| Resolution | 3.0 - 3.2 Å |
| Structure data | EMDB-26999, PDB-8cue: EMDB-27023, PDB-8cw4: |
| Chemicals |  ChemComp-CPL: 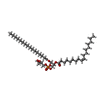 ChemComp-PGW: |
| Source |
|
 Keywords Keywords | STRUCTURAL PROTEIN / conjugation / VirB2 / type IV secretion system / pili / TraM / self transmissable plasmid |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







 Agrobacterium tumefaciens (bacteria)
Agrobacterium tumefaciens (bacteria)