+Search query
-Structure paper
| Title | Structure of S1PR2-heterotrimeric G signaling complex. |
|---|---|
| Journal, issue, pages | Sci Adv, Vol. 8, Issue 13, Page eabn0067, Year 2022 |
| Publish date | Mar 30, 2022 |
 Authors Authors | Hongwen Chen / Kevin Chen / Weijiao Huang / Louis M Staudt / Jason G Cyster / Xiaochun Li /  |
| PubMed Abstract | Sphingosine-1-phosphate (S1P) regulates immune cell trafficking, angiogenesis, and vascular function via its five receptors. Inherited mutations in S1P receptor 2 (S1PR2) occur in individuals with ...Sphingosine-1-phosphate (S1P) regulates immune cell trafficking, angiogenesis, and vascular function via its five receptors. Inherited mutations in S1P receptor 2 (S1PR2) occur in individuals with hearing loss, and acquired mutations in S1PR2 and G occur in a malignant lymphoma. Here, we present the cryo-electron microscopy structure of S1P-bound S1PR2 coupled to the heterotrimeric G. Interaction between S1PR2 intracellular loop 2 (ICL2) and transmembrane helix 4 confines ICL2 to engage the α5 helix of G. Transforming growth factor-α shedding assays and cell migration assays support the key roles of the residues in S1PR2-G complex assembly. The structure illuminates the mechanism of receptor disruption by disease-associated mutations. Unexpectedly, we showed that FTY720-P, an agonist of the other four S1PRs, can trigger G activation via S1PR2. S1PR2 variant can increase the activity of G considerably with FTY720-P and S1P, thus revealing a basis for S1PR drug selectivity. |
 External links External links |  Sci Adv / Sci Adv /  PubMed:35353559 / PubMed:35353559 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 3.19 Å |
| Structure data | EMDB-25712, PDB-7t6b: |
| Chemicals | 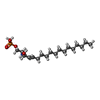 ChemComp-S1P: |
| Source |
|
 Keywords Keywords | SIGNALING PROTEIN/IMMUNE SYSTEM / S1P / S1PR2 / GPCR / membrane protein / cryo-EM / SIGNALING PROTEIN-IMMUNE SYSTEM complex |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 homo sapiens (human)
homo sapiens (human)
