+検索条件
-Structure paper
| タイトル | Cryo-EM structure of the human MT-G signaling complex. |
|---|---|
| ジャーナル・号・ページ | Nat Struct Mol Biol, Vol. 28, Issue 8, Page 694-701, Year 2021 |
| 掲載日 | 2021年8月5日 |
 著者 著者 | Hiroyuki H Okamoto / Hirotake Miyauchi / Asuka Inoue / Francesco Raimondi / Hirokazu Tsujimoto / Tsukasa Kusakizako / Wataru Shihoya / Keitaro Yamashita / Ryoji Suno / Norimichi Nomura / Takuya Kobayashi / So Iwata / Tomohiro Nishizawa / Osamu Nureki /    |
| PubMed 要旨 | Melatonin receptors (MT and MT) transduce inhibitory signaling by melatonin (N-acetyl-5-methoxytryptamine), which is associated with sleep induction and circadian rhythm modulation. Although recently ...Melatonin receptors (MT and MT) transduce inhibitory signaling by melatonin (N-acetyl-5-methoxytryptamine), which is associated with sleep induction and circadian rhythm modulation. Although recently reported crystal structures of ligand-bound MT and MT elucidated the basis of ligand entry and recognition, the ligand-induced MT rearrangement leading to G-coupling remains unclear. Here we report a cryo-EM structure of the human MT-G signaling complex at 3.3 Å resolution, revealing melatonin-induced conformational changes propagated to the G-protein-coupling interface during activation. In contrast to other G-coupled receptors, MT exhibits a large outward movement of TM6, which is considered a specific feature of G-coupled receptors. Structural comparison of G and G complexes demonstrated conformational diversity of the C-terminal entry of the G protein, suggesting loose and variable interactions at the end of the α5 helix of G protein. These notions, together with our biochemical and computational analyses, highlight variable binding modes of Gα and provide the basis for the selectivity of G-protein signaling. |
 リンク リンク |  Nat Struct Mol Biol / Nat Struct Mol Biol /  PubMed:34354246 PubMed:34354246 |
| 手法 | EM (単粒子) |
| 解像度 | 3.3 Å |
| 構造データ | EMDB-30627, PDB-7db6: |
| 化合物 | 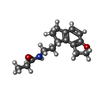 ChemComp-JEV: |
| 由来 |
|
 キーワード キーワード | SIGNALING PROTEIN / gpcr / g-protein / complex |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について





 homo sapiens (ヒト)
homo sapiens (ヒト)
