+検索条件
-Structure paper
| タイトル | Structural basis for activation and allosteric modulation of full-length calcium-sensing receptor. |
|---|---|
| ジャーナル・号・ページ | Sci Adv, Vol. 7, Issue 23, Year 2021 |
| 掲載日 | 2021年6月4日 |
 著者 著者 | Tianlei Wen / Ziyu Wang / Xiaozhe Chen / Yue Ren / Xuhang Lu / Yangfei Xing / Jing Lu / Shenghai Chang / Xing Zhang / Yuequan Shen / Xue Yang /  |
| PubMed 要旨 | Calcium-sensing receptor (CaSR) is a class C G protein-coupled receptor (GPCR) that plays an important role in calcium homeostasis and parathyroid hormone secretion. Here, we present multiple cryo- ...Calcium-sensing receptor (CaSR) is a class C G protein-coupled receptor (GPCR) that plays an important role in calcium homeostasis and parathyroid hormone secretion. Here, we present multiple cryo-electron microscopy structures of full-length CaSR in distinct ligand-bound states. Ligands (Ca and l-tryptophan) bind to the extracellular domain of CaSR and induce large-scale conformational changes, leading to the closure of two heptahelical transmembrane domains (7TMDs) for activation. The positive modulator (evocalcet) and the negative allosteric modulator (NPS-2143) occupy the similar binding pocket in 7TMD. The binding of NPS-2143 causes a considerable rearrangement of two 7TMDs, forming an inactivated TM6/TM6 interface. Moreover, a total of 305 disease-causing missense mutations of CaSR have been mapped to the structure in the active state, creating hotspot maps of five clinical endocrine disorders. Our results provide a structural framework for understanding the activation, allosteric modulation mechanism, and disease therapy for class C GPCRs. |
 リンク リンク |  Sci Adv / Sci Adv /  PubMed:34088669 / PubMed:34088669 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.2 - 7.2 Å |
| 構造データ | EMDB-30644, PDB-7dd5: EMDB-30645, PDB-7dd6:  EMDB-30646: EMDB-30647, PDB-7dd7: |
| 化合物 |  ChemComp-CL:  ChemComp-CA:  ChemComp-NAG: 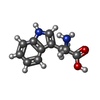 ChemComp-TRP:  ChemComp-YP1:  ChemComp-HOH: 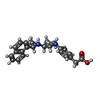 ChemComp-H43: |
| 由来 |
|
 キーワード キーワード | MEMBRANE PROTEIN / Calcium-Sensing Receptor / CaSR / NPS-2143 / active state / Evocalcet |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について










