+検索条件
-Structure paper
| タイトル | Dipeptidyl peptidase 9 sets a threshold for CARD8 inflammasome formation by sequestering its active C-terminal fragment. |
|---|---|
| ジャーナル・号・ページ | Immunity, Vol. 54, Issue 7, Page 1392-1404.e10, Year 2021 |
| 掲載日 | 2021年7月13日 |
 著者 著者 | Humayun Sharif / L Robert Hollingsworth / Andrew R Griswold / Jeffrey C Hsiao / Qinghui Wang / Daniel A Bachovchin / Hao Wu /  |
| PubMed 要旨 | CARD8 detects intracellular danger signals and forms a caspase-1 activating inflammasome. Like the related inflammasome sensor NLRP1, CARD8 autoprocesses into noncovalently associated N-terminal (NT) ...CARD8 detects intracellular danger signals and forms a caspase-1 activating inflammasome. Like the related inflammasome sensor NLRP1, CARD8 autoprocesses into noncovalently associated N-terminal (NT) and C-terminal (CT) fragments and binds the cellular dipeptidyl peptidases DPP8 and 9 (DPP8/9). Certain danger-associated signals, including the DPP8/9 inhibitor Val-boroPro (VbP) and HIV protease, induce proteasome-mediated NT degradation and thereby liberate the inflammasome-forming CT. Here, we report cryoelectron microscopy (cryo-EM) structures of CARD8 bound to DPP9, revealing a repressive ternary complex consisting of DPP9, full-length CARD8, and CARD8-CT. Unlike NLRP1-CT, CARD8-CT does not interact with the DPP8/9 active site and is not directly displaced by VbP. However, larger DPP8/9 active-site probes can directly weaken this complex in vitro, and VbP itself nevertheless appears to disrupt this complex, perhaps indirectly, in cells. Thus, DPP8/9 inhibitors can activate the CARD8 inflammasome by promoting CARD8 NT degradation and by weakening ternary complex stability. |
 リンク リンク |  Immunity / Immunity /  PubMed:34019797 / PubMed:34019797 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.3 - 3.86 Å |
| 構造データ | EMDB-22367, PDB-7jkq: EMDB-22402, PDB-7jn7:  EMDB-22974: |
| 化合物 | 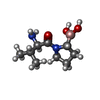 ChemComp-GK2: |
| 由来 |
|
 キーワード キーワード | IMMUNE SYSTEM / HYDROLASE / CARD8 / DPP9 / inflammasome / Val-boroPro (VbP) / talabostat / innate immunity |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 homo sapiens (ヒト)
homo sapiens (ヒト)