+Search query
-Structure paper
| Title | Structural insights into the lipid and ligand regulation of serotonin receptors. |
|---|---|
| Journal, issue, pages | Nature, Vol. 592, Issue 7854, Page 469-473, Year 2021 |
| Publish date | Mar 24, 2021 |
 Authors Authors | Peiyu Xu / Sijie Huang / Huibing Zhang / Chunyou Mao / X Edward Zhou / Xi Cheng / Icaro A Simon / Dan-Dan Shen / Hsin-Yung Yen / Carol V Robinson / Kasper Harpsøe / Bo Svensson / Jia Guo / Hualiang Jiang / David E Gloriam / Karsten Melcher / Yi Jiang / Yan Zhang / H Eric Xu /      |
| PubMed Abstract | Serotonin, or 5-hydroxytryptamine (5-HT), is an important neurotransmitter that activates the largest subtype family of G-protein-coupled receptors. Drugs that target 5-HT, 5-HT, 5-HT and other 5-HT ...Serotonin, or 5-hydroxytryptamine (5-HT), is an important neurotransmitter that activates the largest subtype family of G-protein-coupled receptors. Drugs that target 5-HT, 5-HT, 5-HT and other 5-HT receptors are used to treat numerous disorders. 5-HT receptors have high levels of basal activity and are subject to regulation by lipids, but the structural basis for the lipid regulation and basal activation of these receptors and the pan-agonism of 5-HT remains unclear. Here we report five structures of 5-HT receptor-G-protein complexes: 5-HT in the apo state, bound to 5-HT or bound to the antipsychotic drug aripiprazole; 5-HT bound to 5-HT; and 5-HT in complex with a 5-HT- and 5-HT-selective agonist, BRL-54443. Notably, the phospholipid phosphatidylinositol 4-phosphate is present at the G-protein-5-HT interface, and is able to increase 5-HT-mediated G-protein activity. The receptor transmembrane domain is surrounded by cholesterol molecules-particularly in the case of 5-HT, in which cholesterol molecules are directly involved in shaping the ligand-binding pocket that determines the specificity for aripiprazol. Within the ligand-binding pocket of apo-5-HT are structured water molecules that mimic 5-HT to activate the receptor. Together, our results address a long-standing question of how lipids and water molecules regulate G-protein-coupled receptors, reveal how 5-HT acts as a pan-agonist, and identify the determinants of drug recognition in 5-HT receptors. |
 External links External links |  Nature / Nature /  PubMed:33762731 PubMed:33762731 |
| Methods | EM (single particle) |
| Resolution | 2.9 - 3.1 Å |
| Structure data | EMDB-30971, PDB-7e2x: EMDB-30972, PDB-7e2y: EMDB-30973, PDB-7e2z: EMDB-30974, PDB-7e32: EMDB-30975, PDB-7e33: |
| Chemicals | 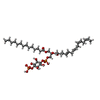 ChemComp-J40:  ChemComp-CLR:  ChemComp-PLM:  ChemComp-HOH: 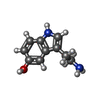 ChemComp-SRO: 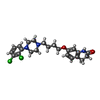 ChemComp-9SC: 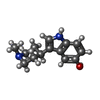 ChemComp-HVU: |
| Source |
|
 Keywords Keywords | SIGNALING PROTEIN / Serotonin / 5-HT1A / GPCR / Complex / G protein / Gi / aripiprazole / 5-HT1D / 5-HT1e / BRL-54443 |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers













 homo sapiens (human)
homo sapiens (human)

