+Search query
-Structure paper
| Title | Immuno- and constitutive proteasome crystal structures reveal differences in substrate and inhibitor specificity. |
|---|---|
| Journal, issue, pages | Cell(Cambridge,Mass. ), Vol. 148, Page 727-738, Year 2012 |
| Publish date | Nov 15, 2011 (structure data deposition date) |
 Authors Authors | Huber, E.M. / Basler, M. / Schwab, R. / Heinemeyer, W. / Kirk, C.J. / Groettrup, M. / Groll, M. |
 External links External links |  Cell(Cambridge,Mass. ) / Cell(Cambridge,Mass. ) /  PubMed:22341445 PubMed:22341445 |
| Methods | X-ray diffraction |
| Resolution | 2.7 - 3.4 Å |
| Structure data |  PDB-3un4:  PDB-3un8:  PDB-3unb:  PDB-3une:  PDB-3unf:  PDB-3unh: |
| Chemicals | 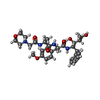 ChemComp-04C:  ChemComp-HOH: 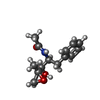 ChemComp-049:  ChemComp-CL:  ChemComp-K:  ChemComp-IOD: |
| Source |
|
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / Proteasome / antigen presentation / drug development / protein degradation / HYDROLASE -HYDROLASE-INHIBITOR complex / HYDROLASE-HYDROLASE INHIBITOR complex / HYDROLASE/HYDROLASE INHIBTIOR / HYDROLASE-HYDROLASE INHIBTIOR complex / 20S proteasome comprises 28 subunits; each subunit adopts the fold of an antiparallel beta-sheet flanked by helices / Protease / Regulatory complexes / Covalent binding of PR-957 to all active sites / HYDROLASE / 20S proteasome comprises 28 subunits / Cytosol |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





