+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
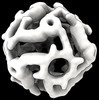
| Title | Computational Designed Nanocage O43_129_+8 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | O43_129_+8 / O43 / 129+2 / 129_+8 / 129 / De Novo Nanocage / DE NOVO PROTEIN | |||||||||
| Biological species | unidentified (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 12.98 Å | |||||||||
 Authors Authors | Weidle C / Kibler RD | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Blueprinting extendable nanomaterials with standardized protein blocks. Authors: Timothy F Huddy / Yang Hsia / Ryan D Kibler / Jinwei Xu / Neville Bethel / Deepesh Nagarajan / Rachel Redler / Philip J Y Leung / Connor Weidle / Alexis Courbet / Erin C Yang / Asim K Bera / ...Authors: Timothy F Huddy / Yang Hsia / Ryan D Kibler / Jinwei Xu / Neville Bethel / Deepesh Nagarajan / Rachel Redler / Philip J Y Leung / Connor Weidle / Alexis Courbet / Erin C Yang / Asim K Bera / Nicolas Coudray / S John Calise / Fatima A Davila-Hernandez / Hannah L Han / Kenneth D Carr / Zhe Li / Ryan McHugh / Gabriella Reggiano / Alex Kang / Banumathi Sankaran / Miles S Dickinson / Brian Coventry / T J Brunette / Yulai Liu / Justas Dauparas / Andrew J Borst / Damian Ekiert / Justin M Kollman / Gira Bhabha / David Baker /   Abstract: A wooden house frame consists of many different lumber pieces, but because of the regularity of these building blocks, the structure can be designed using straightforward geometrical principles. The ...A wooden house frame consists of many different lumber pieces, but because of the regularity of these building blocks, the structure can be designed using straightforward geometrical principles. The design of multicomponent protein assemblies, in comparison, has been much more complex, largely owing to the irregular shapes of protein structures. Here we describe extendable linear, curved and angled protein building blocks, as well as inter-block interactions, that conform to specified geometric standards; assemblies designed using these blocks inherit their extendability and regular interaction surfaces, enabling them to be expanded or contracted by varying the number of modules, and reinforced with secondary struts. Using X-ray crystallography and electron microscopy, we validate nanomaterial designs ranging from simple polygonal and circular oligomers that can be concentrically nested, up to large polyhedral nanocages and unbounded straight 'train track' assemblies with reconfigurable sizes and geometries that can be readily blueprinted. Because of the complexity of protein structures and sequence-structure relationships, it has not previously been possible to build up large protein assemblies by deliberate placement of protein backbones onto a blank three-dimensional canvas; the simplicity and geometric regularity of our design platform now enables construction of protein nanomaterials according to 'back of an envelope' architectural blueprints. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_42031.map.gz emd_42031.map.gz | 102.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-42031-v30.xml emd-42031-v30.xml emd-42031.xml emd-42031.xml | 19.1 KB 19.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_42031_fsc.xml emd_42031_fsc.xml | 12.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_42031.png emd_42031.png | 120.3 KB | ||
| Filedesc metadata |  emd-42031.cif.gz emd-42031.cif.gz | 5.9 KB | ||
| Others |  emd_42031_half_map_1.map.gz emd_42031_half_map_1.map.gz emd_42031_half_map_2.map.gz emd_42031_half_map_2.map.gz | 193.8 MB 193.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-42031 http://ftp.pdbj.org/pub/emdb/structures/EMD-42031 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42031 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-42031 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_42031.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_42031.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.89 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_42031_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_42031_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : O43-129_+8
| Entire | Name: O43-129_+8 |
|---|---|
| Components |
|
-Supramolecule #1: O43-129_+8
| Supramolecule | Name: O43-129_+8 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: A and B chains were expressed separately in e coli. 4 copies of chain A form a side. 3 copies of chain B form a side. The lysate of both expressions are mixed together. 8 sides of each come ...Details: A and B chains were expressed separately in e coli. 4 copies of chain A form a side. 3 copies of chain B form a side. The lysate of both expressions are mixed together. 8 sides of each come together to form a nanoparticle. The nanoparticle is purified by a HIS tag on chain A |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Molecular weight | Theoretical: 2.186 MDa |
-Macromolecule #1: Cage_O43_129_A
| Macromolecule | Name: Cage_O43_129_A / type: other / ID: 1 / Classification: other |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Sequence | String: mGCDAIQAAA ALGEAGISSN EILELLAAAA ELGLDPDAIQ AAAQLGEAGI SSEEIKELLR AAHELGLDPD AIAAAADLGQ AGVSPVEILA LLIAASVLGL DPDAIQAAAA LGEAGISAEE IIELLTAARD LGLDPDAIQA AAQLGEAGIS SEEIKELLRA AHELGLDPDC ...String: mGCDAIQAAA ALGEAGISSN EILELLAAAA ELGLDPDAIQ AAAQLGEAGI SSEEIKELLR AAHELGLDPD AIAAAADLGQ AGVSPVEILA LLIAASVLGL DPDAIQAAAA LGEAGISAEE IIELLTAARD LGLDPDAIQA AAQLGEAGIS SEEIKELLRA AHELGLDPDC IAAAADLGQA GISSSEITAL LLAAAAIELA KRADDKDVRE IVRDALELAS RSTNDEVIRL ALEAAVLAAR STDSDVLEIV KDALELAKQS TNEEVIKLAL KAAVLAAKST DEEVLEEVKE ALRRAKESTD EEEIKEELRK AVEEAEGGSW lehhhhhh |
| Recombinant expression | Organism:  |
-Macromolecule #2: cage_O43_129_+8_B
| Macromolecule | Name: cage_O43_129_+8_B / type: other / ID: 2 / Classification: other |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Sequence | String: mGDDLLLKLL ELLVEQARVS AEFARRQGDE KMLEEVARKA EEVARKAEEI ARKARKEGNL ELALKALEIL VRAAHVLAEI ARERGNEELQ KKAHKLAKEA LRQVIEIAIR AIQEGNLELA IIALHISVRI AEVLLETRPD DREEIREQQA IFELLIAALE AAIRLEKLKE ...String: mGDDLLLKLL ELLVEQARVS AEFARRQGDE KMLEEVARKA EEVARKAEEI ARKARKEGNL ELALKALEIL VRAAHVLAEI ARERGNEELQ KKAHKLAKEA LRQVIEIAIR AIQEGNLELA IIALHISVRI AEVLLETRPD DREEIREQQA IFELLIAALE AAIRLEKLKE EGAPPEQIER VAEHGLERLK EIAKEISKEV DSPESKRIAY KIVAAAAEFL LKILAEGGAT PEQLERVTEH ALEVLKEVAK ELADSPESVR EAVRLISKLT QEGLKQLKEI GAPPEQIERV AEHGLEVLKE IAKYGSKLTD SPELKRELYR IISETAKELL KILAEGGATP EQLERVTKHA LEVLKEVAKE LADSPESVRE AVRLISKLTQ EGLKQLKEIG APPEQIERVA EHGLEVLKEI AKYGSKLTDS PELKRELYRI ISETAKELLK ILAEGGATPE QLERVTKHAL EVLKEVAKEL ADSPESGLAA LAAIASLAKL GLEQLKEIGA PPEQQRRVTK AGIEAVREIY RYGRKLY |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.8 mg/mL |
|---|---|
| Buffer | pH: 8 / Component - Concentration: 300.0 mM / Component - Formula: NaCl / Component - Name: sodium chloride / Details: 25mM Tris, 300mM NaCl |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS GLACIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number grids imaged: 1 / Average exposure time: 5.0 sec. / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.8 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Initial model type: in silico model |
|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
 Movie
Movie Controller
Controller























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)