+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of human Wnt7a bound to WLS and RECK | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | SIGNALING PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of establishment of blood-brain barrier / negative regulation of metalloendopeptidase activity / Wnt protein secretion / blood vessel maturation / positive regulation of Wnt protein secretion / WNT ligand biogenesis and trafficking / Post-translational modification: synthesis of GPI-anchored proteins / metalloendopeptidase inhibitor activity / cementum mineralization / hindbrain development ...regulation of establishment of blood-brain barrier / negative regulation of metalloendopeptidase activity / Wnt protein secretion / blood vessel maturation / positive regulation of Wnt protein secretion / WNT ligand biogenesis and trafficking / Post-translational modification: synthesis of GPI-anchored proteins / metalloendopeptidase inhibitor activity / cementum mineralization / hindbrain development / regulation of extracellular matrix organization / Wnt-protein binding / exocrine pancreas development / embryonic forelimb morphogenesis / Wnt signalosome / sprouting angiogenesis / anterior/posterior axis specification / midbrain development / endopeptidase inhibitor activity / organelle membrane / positive regulation of Wnt signaling pathway / mesoderm formation / endomembrane system / regulation of angiogenesis / canonical Wnt signaling pathway / coreceptor activity / side of membrane / embryo implantation / extracellular matrix organization / negative regulation of cell migration / intracellular protein transport / serine-type endopeptidase inhibitor activity / trans-Golgi network / Wnt signaling pathway / endocytic vesicle membrane / positive regulation of canonical Wnt signaling pathway / early endosome membrane / cytoplasmic vesicle / positive regulation of canonical NF-kappaB signal transduction / early endosome / Golgi membrane / endoplasmic reticulum membrane / Golgi apparatus / endoplasmic reticulum / extracellular exosome / extracellular region / identical protein binding / membrane / plasma membrane / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.23 Å | ||||||||||||
 Authors Authors | Qi X / Hu Q / Li X | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Cell / Year: 2023 Journal: Cell / Year: 2023Title: Molecular basis of Wnt biogenesis, secretion, and Wnt7-specific signaling. Authors: Xiaofeng Qi / Qinli Hu / Nadia Elghobashi-Meinhardt / Tao Long / Hongwen Chen / Xiaochun Li /   Abstract: Wnt proteins are enzymatically lipidated by Porcupine (PORCN) in the ER and bind to Wntless (WLS) for intracellular transport and secretion. Mechanisms governing the transfer of these low-solubility ...Wnt proteins are enzymatically lipidated by Porcupine (PORCN) in the ER and bind to Wntless (WLS) for intracellular transport and secretion. Mechanisms governing the transfer of these low-solubility Wnts from the ER to the extracellular space remain unclear. Through structural and functional analyses of Wnt7a, a crucial Wnt involved in central nervous system angiogenesis and blood-brain barrier maintenance, we have elucidated the principles of Wnt biogenesis and Wnt7-specific signaling. The Wnt7a-WLS complex binds to calreticulin (CALR), revealing that CALR functions as a chaperone to facilitate Wnt transfer from PORCN to WLS during Wnt biogenesis. Our structures, functional analyses, and molecular dynamics simulations demonstrate that a phospholipid in the core of Wnt-bound WLS regulates the association and dissociation between Wnt and WLS, suggesting a lipid-mediated Wnt secretion mechanism. Finally, the structure of Wnt7a bound to RECK, a cell-surface Wnt7 co-receptor, reveals how RECK engages the N-terminal domain of Wnt7a to activate Wnt7-specific signaling. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41765.map.gz emd_41765.map.gz | 117.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41765-v30.xml emd-41765-v30.xml emd-41765.xml emd-41765.xml | 17.7 KB 17.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41765.png emd_41765.png | 48.2 KB | ||
| Filedesc metadata |  emd-41765.cif.gz emd-41765.cif.gz | 6.5 KB | ||
| Others |  emd_41765_half_map_1.map.gz emd_41765_half_map_1.map.gz emd_41765_half_map_2.map.gz emd_41765_half_map_2.map.gz | 116 MB 116 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41765 http://ftp.pdbj.org/pub/emdb/structures/EMD-41765 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41765 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41765 | HTTPS FTP |
-Validation report
| Summary document |  emd_41765_validation.pdf.gz emd_41765_validation.pdf.gz | 998.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41765_full_validation.pdf.gz emd_41765_full_validation.pdf.gz | 998.1 KB | Display | |
| Data in XML |  emd_41765_validation.xml.gz emd_41765_validation.xml.gz | 14 KB | Display | |
| Data in CIF |  emd_41765_validation.cif.gz emd_41765_validation.cif.gz | 16.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41765 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41765 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41765 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41765 | HTTPS FTP |
-Related structure data
| Related structure data |  8tzpMC  8tzoC  8tzsC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41765.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41765.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.83 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #1
| File | emd_41765_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
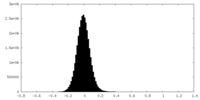
| Density Histograms |
-Half map: #2
| File | emd_41765_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
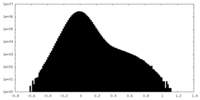
| Density Histograms |
- Sample components
Sample components
-Entire : Wnt7a-WLS-RECK Complex
| Entire | Name: Wnt7a-WLS-RECK Complex |
|---|---|
| Components |
|
-Supramolecule #1: Wnt7a-WLS-RECK Complex
| Supramolecule | Name: Wnt7a-WLS-RECK Complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Protein Wnt-7a
| Macromolecule | Name: Protein Wnt-7a / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 39.062977 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MNRKARRCLG HLFLSLGMVY LRIGGFSSVV ALGASIICNK IPGLAPRQRA ICQSRPDAII VIGEGSQMGL DECQFQFRNG RWNCSALGE RTVFGKELKV GSREAAFTYA IIAAGVAHAI TAACTQGNLS DCGCDKEKQG QYHRDEGWKW GGCSADIRYG I GFAKVFVD ...String: MNRKARRCLG HLFLSLGMVY LRIGGFSSVV ALGASIICNK IPGLAPRQRA ICQSRPDAII VIGEGSQMGL DECQFQFRNG RWNCSALGE RTVFGKELKV GSREAAFTYA IIAAGVAHAI TAACTQGNLS DCGCDKEKQG QYHRDEGWKW GGCSADIRYG I GFAKVFVD AREIKQNART LMNLHNNEAG RKILEENMKL ECKCHGVSGS CTTKTCWTTL PQFRELGYVL KDKYNEAVHV EP VRASRNK RPTFLKIKKP LSYRKPMDTD LVYIEKSPNY CEEDPVTGSV GTQGRACNKT APQASGCDLM CCGRGYNTHQ YAR VWQCNC KFHWCCYVKC NTCSERTEMY TCK UniProtKB: UNIPROTKB: O00755 |
-Macromolecule #2: Protein wntless homolog
| Macromolecule | Name: Protein wntless homolog / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 62.317973 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MAGAIIENMS TKKLCIVGGI LLVFQIIAFL VGGLIAPGPT TAVSYMSVKC VDARKNHHKT KWFVPWGPNH CDKIRDIEEA IPREIEAND IVFSVHIPLP HMEMSPWFQF MLFILQLDIA FKLNNQIREN AEVSMDVSLA YRDDAFAEWT EMAHERVPRK L KCTFTSPK ...String: MAGAIIENMS TKKLCIVGGI LLVFQIIAFL VGGLIAPGPT TAVSYMSVKC VDARKNHHKT KWFVPWGPNH CDKIRDIEEA IPREIEAND IVFSVHIPLP HMEMSPWFQF MLFILQLDIA FKLNNQIREN AEVSMDVSLA YRDDAFAEWT EMAHERVPRK L KCTFTSPK TPEHEGRYYE CDVLPFMEIG SVAHKFYLLN IRLPVNEKKK INVGIGEIKD IRLVGIHQNG GFTKVWFAMK TF LTPSIFI IMVWYWRRIT MMSRPPVLLE KVIFALGISM TFINIPVEWF SIGFDWTWML LFGDIRQGIF YAMLLSFWII FCG EHMMDQ HERNHIAGYW KQVGPIAVGS FCLFIFDMCE RGVQLTNPFY SIWTTDIGTE LAMAFIIVAG ICLCLYFLFL CFMV FQVFR NISGKQSSLP AMSKVRRLHY EGLIFRFKFL MLITLACAAM TVIFFIVSQV TEGHWKWGGV TVQVNSAFFT GIYGM WNLY VFALMFLYAP SHKNYGEDQS NGDLGVHSGE ELQLTTTITH VDGPTEIYKL TRKEAQE UniProtKB: Protein wntless homolog |
-Macromolecule #3: Reversion-inducing cysteine-rich protein with Kazal motifs
| Macromolecule | Name: Reversion-inducing cysteine-rich protein with Kazal motifs type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 106.573461 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MATVRASLRG ALLLLLAVAG VAEVAGGLAP GSAGALCCNH SKDNQMCRDV CEQIFSSKSE SRLKHLLQRA PDYCPETMVE IWNCMNSSL PGVFKKSDGW VGLGCCELAI ALECRQACKQ ASSKNDISKV CRKEYENALF SCISRNEMGS VCCSYAGHHT N CREYCQAI ...String: MATVRASLRG ALLLLLAVAG VAEVAGGLAP GSAGALCCNH SKDNQMCRDV CEQIFSSKSE SRLKHLLQRA PDYCPETMVE IWNCMNSSL PGVFKKSDGW VGLGCCELAI ALECRQACKQ ASSKNDISKV CRKEYENALF SCISRNEMGS VCCSYAGHHT N CREYCQAI FRTDSSPGPS QIKAVENYCA SISPQLIHCV NNYTQSYPMR NPTDSLYCCD RAEDHACQNA CKRILMSKKT EM EIVDGLI EGCKTQPLPQ DPLWQCFLES SQSVHPGVTV HPPPSTGLDG AKLHCCSKAN TSTCRELCTK LYSMSWGNTQ SWQ EFDRFC EYNPVEVSML TCLADVREPC QLGCRNLTYC TNFNNRPTEL FRSCNAQSDQ GAMNDMKLWE KGSIKMPFIN IPVL DIKKC QPEMWKAIAC SLQIKPCHSK SRGSIICKSD CVEILKKCGD QNKFPEDHTA ESICELLSPT DDLKNCIPLD TYLRP STLG NIVEEVTHPC NPNPCPANEL CEVNRKGCPS GDPCLPYFCV QGCKLGEASD FIVRQGTLIQ VPSSAGEVGC YKICSC GQS GLLENCMEMH CIDLQKSCIV GGKRKSHGTS FSIDCNVCSC FAGNLVCSTR LCLSEHSSED DRRTFTGLPC NCADQFV PV CGQNGRTYPS ACIARCVGLQ DHQFEFGSCM SKDPCNPNPC QKNQRCIPKP QVCLTTFDKF GCSQYECVPR QLACDQVQ D PVCDTDHMEH NNLCTLYQRG KSLSYKGPCQ PFCRATEPVC GHNGETYSSV CAAYSDRVAV DYYGDCQAVG VLSEHSSVA ECASVKCPSL LAAGCKPIIP PGACCPLCAG MLRVLFDKEK LDTIAKVTNK KPITVLEILQ KIRMHVSVPQ CDVFGYFSIE SEIVILIIP VDHYPKALQI EACNKEAEKI ESLINSDSPT LASHVPLSAL IISQVQVSSS VPSAGVRARP SCHSLLLPLS L GLALHLLW TYN UniProtKB: Reversion-inducing cysteine-rich protein with Kazal motifs |
-Macromolecule #4: PALMITOLEIC ACID
| Macromolecule | Name: PALMITOLEIC ACID / type: ligand / ID: 4 / Number of copies: 1 / Formula: PAM |
|---|---|
| Molecular weight | Theoretical: 254.408 Da |
| Chemical component information | 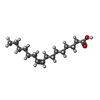 ChemComp-PAM: |
-Macromolecule #5: (2S)-3-(hexadecanoyloxy)-2-[(9Z)-octadec-9-enoyloxy]propyl 2-(tri...
| Macromolecule | Name: (2S)-3-(hexadecanoyloxy)-2-[(9Z)-octadec-9-enoyloxy]propyl 2-(trimethylammonio)ethyl phosphate type: ligand / ID: 5 / Number of copies: 1 / Formula: POV |
|---|---|
| Molecular weight | Theoretical: 760.076 Da |
| Chemical component information |  ChemComp-POV: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.23 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 87543 |
| Initial angle assignment | Type: NOT APPLICABLE |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller







 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)